International Journal of Agricultural Science and Food Technology
Gene pool, classification and its importance in modern crop improvement program
1Ethiopian Institute of Agricultural Research, Chiro National Sorghum Research and Training Center, Chiro, Ethiopia
2Ethiopian Institute of Agricultural Research, Bako National Maize Research Center, Bako, Ethiopia
Author and article information
Cite this as
Yali W, Mitiku T (2024) Gene pool, classification and its importance in modern crop improvement program. Int J Agric Sc Food Technol. 2024; 10(2): 68-73. Available from: 10.17352/2455-815X.000209
Copyright License
© 2024 Yali W, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Alleles present in a population at a given period are referred to as the gene pool. It is the total amount and variety of genes and alleles available for transmission to the next generation in a sexually reproducing population. In comparison to formal taxonomy, Harlan and Wet identified types of gene pools as main, secondary, tertiary, and quaternary gene pools for the classification of each crop and its related species. The total quantity of genes, types and variants of genes, proportion, and distribution of genes are all revealed by studying the gene pool of a population. Selection, mutation, gene flow, and genetic drift are evolutionary mechanisms that influence a population’s gene pool. The number of genes and the number of people who carry these genes determine the size of the gene pool. Because of chance events, the number of each gene in a gene pool might shift over time. As a result, the goal of this review study is to evaluate the gene pool, classification, and importance of the gene pool in modern crop development programs.
All the genes and alleles present in all such individuals, which hybridize or can hybridize with each other, make up the gene pool [1]. It is the collection of various genes in a population of a specific species at a specific time. The term “gene pool” is most commonly used to refer to a group of individuals belonging to a single species, and the term “gene pool” refers to all of the genes and gene combinations present in the population [2]. The term “gene pool” refers to the total number of alleles of genes found at all loci within a single species population. Thus, the phrase “gene pool” is used to define the collection of all genes or genetic information found in any population of a specific species [3].
A gene pool is the total quantity and variety of genes and alleles available for transmission to the next generation in a sexually reproducing population [4]. Plant breeders benefit from the gene pool concept because it helps them choose germplasm to employ in hybridizations for plant improvement. A vast gene pool shows a high level of genetic variety, which is linked to populations that can withstand periods of strong selection [5]. Crop improvement initiatives rely on accessible genetic diversity for specific traits in the primary and, if appropriate, secondary gene pools of a given crop species for breeding, selection, and agronomic evaluation [6].
Plant breeders can construct dynamic gene pools from which they can draw materials for crop improvement by using a method of introgression (crossing and backcrossing selected entrants with desired traits into adapted stocks) and incorporation [7]. These researchers advised that the search for desirable genes begins with materials from the primary gene pool (related species), then goes on to the secondary gene pool, and finally to the tertiary gene pool if necessary [8]. Then, using the universal gene pool, proprietary germplasm, and genetic technologies, plant geneticists create new hybrids [9]. The overall goal of this review study is to evaluate the gene pool, kinds, and importance of these genes in modern crop development programs.
Literature review
Concept of gene pool: The notion of a gene pool was developed by Russian biologist Alexander Sergeevich Serebrovsky, who invented the term ‘genofond’, which means “gene pool” in English. The gene pool refers to a significant collection of genetic variety within a single species [10]. This refers to all of the alleles found in a population of a certain species at a given locus. The gene pool definition also includes all genes, regardless of expression, i.e., whether they are expressed or not, they are all included in the gene pool definition [11]. When a population is robust and can withstand natural selection processes, the gene pool is usually regarded [12].
The concept of biological fitness is another concept that is tied to the concept of gene pool. Biological fitness is determined by a species’s gene pool’s ability to withstand numerous changes that can result in intense natural selection [13]. If a species is unable to adapt to changing conditions in an ecosystem, it may become extinct. Biological fitness is a term used to describe a species’ ability to adapt to changes [14]. The biological fitness of a species is considered to be high if the gene pool contains a lot of diversity in alleles, whereas it is said to be low if the gene pool contains little diversity [15]. Thus, high genetic diversity in the gene pool leads to high fitness, whereas low genetic diversity resulting from inbreeding or bottleneck events can lead to decreased biological fitness and higher odds of extinction [16].
There are several situations in which a species can persist even with low genetic diversity, i.e. low biological fitness, if the fitness is raised by processes such as genetic drift [17]. For a given species, genetic drift is defined as a shift in the frequency of a specific gene variant, or allele. If new genetic variants more adapted to changing environments are introduced into the population of the species as a result of these changes, there is a potential to boost the species’ biological fitness even if the gene pool variety is low [18]. Thus, the gene pool does not reflect biological fitness in and of itself but rather is an indicator of biological diversity that provides relevant information about the fitness of a species population [19].
Changes in the gene pool: Small changes in the amount of each type of gene can occur throughout generations for a variety of reasons [18]. The principal causes of changes in a population’s gene pool include gene mutation, gene flow, genetic drift, and natural selection [20]. The gene pool does not always remain consistent [21]. Changes in the gene pool can lead to changes in the genetic diversity of the species population [22]. The content of the gene pool can alter over time as a result of evolutionary processes. Changes in the makeup of the gene pool can be caused by a variety of factors, including mutation, natural selection, and genetic drift [23]. These adjustments are necessary for any population of animals to survive as the environment changes. These genetic variants provide a diversified gene pool, allowing people in the community to adapt to changing environments. Changes in the environment resulted in genetic alterations that became part of the human gene pool in that location [24].
The diversity of alleles present in a population is reflected in the gene pool, which is an indicator of biological fitness. The ability of a population of a species to adapt to a specific environment and evolve is affected in part by the gene pool [25]. These modifications may aid in the diversification of the gene pool in question. However, this does not imply that genetic variety and gene pool have the same meaning in a statement. Genetic diversity is a trait that emerges from a variety of events that lead to the formation of a population of organisms. In a population that makes up a gene pool, it can have only one genetic variation of a gene or numerous alleles. As a result, it should be highlighted that each is a component that indicates the presence of another [26].
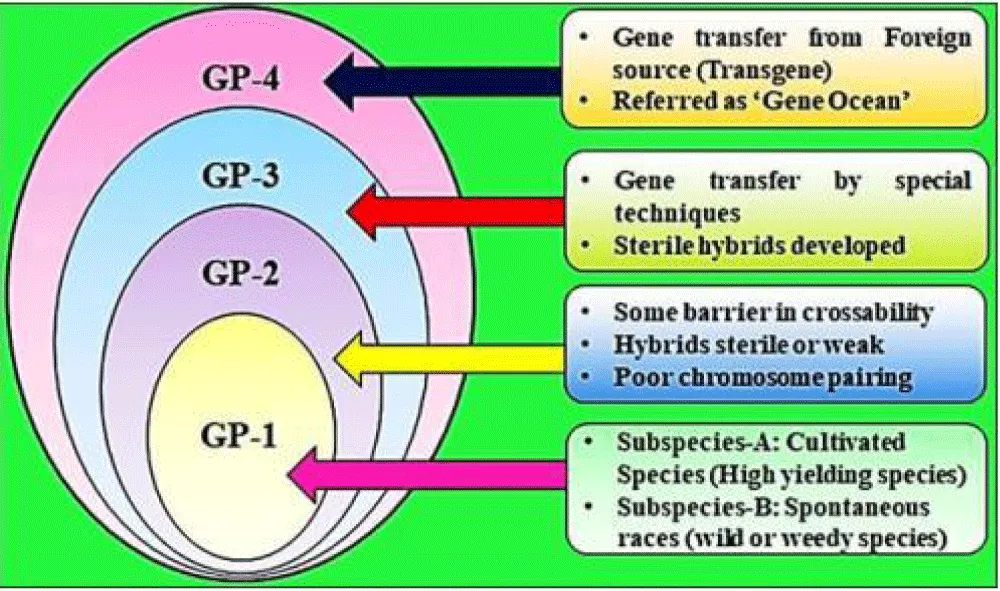
Types of gene pools in crop breeding: The total genetic variety in a species’ breeding population and closely related species capable of crossing with it is referred to as the gene pool. In comparison to formal taxonomy, Harlan and Wet developed categories of gene pools for the classification of each crop and its related species [27].
Primary gene pool: Gene Pool one (GP 1) is another name for the primary gene pool [28]. This pool’s inhabitants are all of the same species and can freely mate. This gene pool is unique in that it is easy to cross, resulting in fertile hybrids with strong chromosome pairing and proper gene segregation, allowing for easy gene transfer [29]. Total genetic variety in a crop’s breeding population and closely related species with whom gene transfer is straightforward since they frequently interbreed or can be crossed. The major gene pool is the one in which intermating is simple and results in viable hybrids [30]. Plants of the same species or closely related species that intermate produce viable progeny are included. Genes can be exchanged across lines in such a gene pool simply by doing normal crosses with normal seed sets, segregation, and recombination [31].
Secondary gene pool: Gene Pool two (GP 2) is a secondary gene pool [32]. Total genetic variation in a population of less closely related species from which gene transfer to the crop is possible but difficult to achieve using traditional breeding procedures [33]. Due to several issues with F1 infertility, gene transfer, and combination may face challenges. This gene pool is made up of species that are not identical but are more closely connected to one of the basic gene pools [34]. Hybrids can be produced, although they may be sterile (i.e., have lower fertility), and chromosome pairing and gene segregation may be poor. Nonetheless, it can generate a wide range of hybrids. The secondary gene pool is the genetic material that results in partial fertility when crossed with GP 1. It contains plants from related species that have certain obstacles to cross-pollination with crops [35]. Such materials can be mated with the primary gene pool, but the hybrids are usually infertile or weak, with some fertile progeny. The GP2 of most crop plants is mostly made up of species from the same genus [36].
Tertiary gene pool: This is also known as the third gene pool (GP 3). Total genetic variation in a population of a more distant species, from which gene transfer to the crop is unfeasible or, if achievable, necessitates somatic fusion or in-vitro procedures such as embryo rescue for F1 generation and survival [37]. F1 sterility is a major impediment to gene transfer. This pool contains species that are completely different from the gene pool under discussion, and crossing them will produce extremely few if any, hybrids. This is the shallow end of the gene pool, so to speak [38]. The genetic material that results in sterile hybrids when crossed with the primary gene pool is referred known as the tertiary gene pool. It contains components that can be crossed with GP 1, however the hybrids are either fatal or sterile as a result of aberrant embryo development [39]. Special techniques like as embryo culture, tissue culture, chromosomal doubling, or the use of bridge species (with members of secondary gene pools) can be used to transfer genes from such material to the primary gene pool [40].
Quaternary gene pool (GP4): Any synthetic strains having nucleic acid frequencies (DNA or RNA) that do not occur in nature should be included in the fourth gene pool [41]. The recent advancement in genetic engineering technology has brought the entire world of living organisms within reach of any crop plant’s germplasm. The enormous genetic diversity results from unrelated plant species or other organisms [42]. Pollination and tissue culture procedures are unable to transmit genes, necessitating the use of genetic engineering techniques. Through recombinant DNA technology, genes can now be transferred not only between species or genera but also between other types of organisms [43]. The ‘Bt’ gene (Transgene) was transferred from a bacteria (Bacillus thuringiensis) to cotton, maize, tomato, and other plant species, expanding the gene pool beyond human imagination. Modern science has thus made available a new form of gene pool (GP-4) that might be referred to as a “Gene Ocean” [44] Figure 1.
Gene pool centers
Gene pool centers are locations around the world where important crop plants and domestic animals made their first appearance. They have a diverse collection of wild counterparts to cultivated plant species as well as useful tropical plants [45]. Gene pool centers also house a variety of warm and temperate zone species. It denotes the total genetic diversity present in a population or species [46]. A vast gene pool denotes a high level of genetic variety, which is linked to strong populations that can withstand periods of harsh selection. Low genetic variety, on the other hand, can result in decreased biological fitness and an increased risk of extinction [47]. A geographical place where a group of species, either domesticated or wild, first formed their distinguishing features is known as the center of origin [48].
Genetic erosion
Natural or man-made processes that result in a loss of genetic diversity. Genetic erosion is the loss of genetic variety in farmed or natural populations as a result of human intervention [49]. Natural forces, as well as the acts of crop producers, plant breeders, curators of germplasm banks, and others in society, can cause genetic variation loss. Genetic erosion is a term used to explain the loss of variety in crops as a result of agricultural modernization. At three stages of integration, genetic degradation can occur viz., crop, variety, and allele [50].
The introduction of newer cultivars has been linked to genetic degradation in crops. Genetic erosion in native plant populations is caused by habitat loss and fragmentation, but it can also be caused by a limited genetic foundation in the original collections or by techniques that diminish genetic variety [51]. Genetic erosion can refer to the disappearance of genetically distinct populations, the loss or change in frequency of certain alleles (different forms of a gene) within populations or between species, or the disappearance of allele combinations [52].
Major losses of habitat and resident plant populations; habitat fragmentation; management activities such as thinning, harvesting, or nursery selections that target specific features of plants; and planting material from a narrow genetic collection in revegetation efforts are all factors that could contribute to genetic erosion in native plant species [53]. Genetic erosion is the loss of genetic variability owing to a variety of circumstances over some time and in a specific location. Individual genes or combinations of genes can be lost. The decrease in genetic variety over time is referred to as genetic loss [54]. Genetic loss is mostly due to agricultural modernization, which includes the replacement of landraces with new improved kinds. Plant breeding initiatives are being hampered by the loss of genetic variety [55].
Genetic vulnerability
Genetic vulnerability is a word used to describe a group of plants’ genetic homogeneity and uniformity, which makes them susceptible to a large-scale pest, pathogen, or environmental danger [56]. It’s a big deal in current plant breeding, and it’s largely due to the way breeders go about generating new and better cultivars for modern civilization. Crop evolution, breeding trends, biological technology trends, crop producer decisions, consumer demands and tastes, and other factors are all elements that contribute to genetic susceptibility [57]. A specific crop cultivar (genotype) is developed for the agricultural production system as a consequence of a combination of the above elements. Using varied and unrelated parents in breeding programs, as well as unrelated causes of male sterility, semi-dwarfism, and other genetic vulnerabilities, can be avoided [58].
Role of gene pool in plant breeding
Plant breeders face multiple global challenges that affect food security, productivity, accessibility, and nutritional quality [59]. Gene Pool serves as a guide for planning breeding activities. The effective utilization of the pant gene pool would contribute to solving constraints that limit crop productivity. The identification of the crop gene pool, which comprises farmed forms of the crop species, wild species of the crop, and the crop’s progenitors, is an important goal in plant breeding [60]. Closely related species are also included in the crop gene pool, even if they did not act as direct progenitors. The gene pool concept is mostly used to demarcate taxa and determine levels of interfertility between these taxa and the associated crop [61].
Plant breeders benefit from the gene pool concept because it helps them choose germplasm to employ in hybridizations for plant improvement. Each gene serves a specific purpose, such as conferring a specific trait to the plant/animal, disease resistance, climatic tolerance, and so on [62]. As a result, a population with more genetic variety will be better prepared to deal with disease outbreaks or drastic environmental changes, as they will be more likely to possess genes that protect them from such threats. Populations with a smaller number of genes in their gene pool, on the other hand, will be more vulnerable to such issues, which could lead to them being endangered or even extinct.
As a result, communities with a wide gene pool have a better chance of surviving, whereas populations with small gene pools are at risk of developing genetic disorders, abnormalities, and infertility [63]. Farmers globally lost over 75% of agricultural genetic variety as they moved to genetically uniform, high-yielding cultivars and abandoned multiple local types. However, having access to genetic material is critical for adapting and improving agriculture in the face of risks like as disease or a warming climate that can change growing conditions [64]. Plant breeders are now using those genes to create disease-resistant wheat cultivars [58].
Conclusion
The total amount of genes found in a community is referred to as the gene pool; populations with larger gene pools have more genes and thus higher genetic diversity. For long-term development, maintaining the gene pool is crucial. The most frequent breeding methods used in productivity enhancement include gene pool enrichment via native or alien germplasm, selection, and hybridization. Crop improvement efforts, both traditional and nontraditional, should focus on closing the yield gap between potential and realized yields by removing significant production limitations.
- Majhi PK. Chapter-4 Gene Pool Concept in Plant Breeding. In: Current Research and Innovations in Plant Pathology. 2020; 85.
- Baker RJ, Bradley RD. SPECIATION IN MAMMALS AND THE GENETIC SPECIES CONCEPT. J Mammal. 2006 Aug 1;87(4):643-662. doi: 10.1644/06-MAMM-F-038R2.1. PMID: 19890476; PMCID: PMC2771874.
- Price PW. Evolutionary Biology of Parasites. (MPB-15), Volume 15. Princeton University Press; 2020.
- Burke MK. How does adaptation sweep through the genome? Insights from long-term selection experiments. Proc Biol Sci. 2012; 279(1749):5029-5038.
- Hill CB, Angessa TT, Zhang XQ, Chen K, Zhou G, Tan C, Wang P, Westcott S, Li C. A global barley panel revealing genomic signatures of breeding in modern Australian cultivars. Plant J. 2021 Apr;106(2):419-434. doi: 10.1111/tpj.15173. Epub 2021 Mar 9. PMID: 33506596.
- Heffner EL, Sorrells ME, Jannink JL. Genomic selection for crop improvement. Crop Sci. 2009; 49(1):1-12.
- Ortiz R, Braun HJ, Crossa J. Wheat genetic resources enhancement by the International Maize and Wheat Improvement Center (CIMMYT). Genet Resour Crop Evol. 2008; 55(7):1095-1140.
- Jones TA. The restoration gene pool concept: beyond the native versus non‐native debate. Restor Ecol. 2003;11(3):281-290.
- Kloppenburg J. Re-purposing the master's tools: the open source seed initiative and the struggle for seed sovereignty. J Peasant Stud. 2014;41(6):1225-1246.
- Kaewwongwal A, Kongjaimun A, Somta P, Chankaew S, Yimram T, Srinives P. Genetic diversity of the black gram [Vigna mungo (L.) Hepper] gene pool as revealed by SSR markers. Breed Sci. 2015 Mar;65(2):127-37. doi: 10.1270/jsbbs.65.127. Epub 2015 Mar 1. PMID: 26069442; PMCID: PMC4430507.
- Ye Y, Dai Q, Qi H. A novel defined pyroptosis-related gene signature for predicting the prognosis of ovarian cancer. Cell Death Discov. 2021 Apr 7;7(1):71. doi: 10.1038/s41420-021-00451-x. PMID: 33828074; PMCID: PMC8026591.
- Exposito-Alonso M; 500 Genomes Field Experiment Team; Burbano HA, Bossdorf O, Nielsen R, Weigel D. Natural selection on the Arabidopsis thaliana genome in present and future climates. Nature. 2019 Sep;573(7772):126-129. doi: 10.1038/s41586-019-1520-9. Epub 2019 Aug 28. Erratum in: Nature. 2019 Sep 30;: PMID: 31462776.
- Waldvogel AM, Feldmeyer B, Rolshausen G, Exposito-Alonso M, Rellstab C, Kofler R, Mock T, Schmid K, Schmitt I, Bataillon T, Savolainen O, Bergland A, Flatt T, Guillaume F, Pfenninger M. Evolutionary genomics can improve prediction of species' responses to climate change. Evol Lett. 2020 Jan 14;4(1):4-18. doi: 10.1002/evl3.154. PMID: 32055407; PMCID: PMC7006467.
- Giangrande A, Pierri C, Del Pasqua M, Gravili C, Gambi MC, Gravina MF. The Mediterranean in check: Biological invasions in a changing sea. Mar Ecol. 2020; 41(2)
- Vandewoestijne S, Schtickzelle N, Baguette M. Positive correlation between genetic diversity and fitness in a large, well-connected metapopulation. BMC Biol. 2008 Nov 5;6:46. doi: 10.1186/1741-7007-6-46. PMID: 18986515; PMCID: PMC2587462.
- Szczecińska M, Sramko G, Wołosz K, Sawicki J. Genetic Diversity and Population Structure of the Rare and Endangered Plant Species Pulsatilla patens (L.) Mill in East Central Europe. PLoS One. 2016 Mar 22;11(3):e0151730. doi: 10.1371/journal.pone.0151730. PMID: 27003296; PMCID: PMC4803199.
- Bickham JW, Sandhu S, Hebert PD, Chikhi L, Athwal R. Effects of chemical contaminants on genetic diversity in natural populations: implications for biomonitoring and ecotoxicology. Mutat Res. 2000 Jul;463(1):33-51. doi: 10.1016/s1383-5742(00)00004-1. PMID: 10838208.
- Slatkin M. Gene flow and the geographic structure of natural populations. Science. 1987 May 15;236(4803):787-92. doi: 10.1126/science.3576198. PMID: 3576198.
- Hughes AR, Inouye BD, Johnson MT, Underwood N, Vellend M. Ecological consequences of genetic diversity. Ecol Lett. 2008 Jun;11(6):609-23. doi: 10.1111/j.1461-0248.2008.01179.x. Epub 2008 Apr 8. PMID: 18400018.
- Clark MA, Choi J, Douglas M. 24.2–Population Genetics. Biology 110 PSU Dubois. 2018.
- Luong NH, La Poutré H, Bosman PA. Multi-objective gene-pool optimal mixing evolutionary algorithm with the interleaved multi-start scheme. Swarm Evol Comput. 2018;40:238-254.
- Hoban S, Bruford M, Jackson JDU, Lopes-Fernandes M, Heuertz M, Hohenlohe PA. Genetic diversity targets and indicators in the CBD post-2020 Global Biodiversity Framework must be improved. Biol Conserv. 2020;248:108654.
- Johnson MTJ, Prashad CM, Lavoignat M, Saini HS. Contrasting the effects of natural selection, genetic drift and gene flow on urban evolution in white clover ( Trifolium repens ). Proc Biol Sci. 2018 Jul 18;285(1883):20181019. doi: 10.1098/rspb.2018.1019. PMID: 30051843; PMCID: PMC6083247.
- Smith AL, Hodkinson TR, Villellas J, Catford JA, Csergő AM, Blomberg SP, Crone EE, Ehrlén J, Garcia MB, Laine AL, Roach DA, Salguero-Gómez R, Wardle GM, Childs DZ, Elderd BD, Finn A, Munné-Bosch S, Baudraz MEA, Bódis J, Brearley FQ, Bucharova A, Caruso CM, Duncan RP, Dwyer JM, Gooden B, Groenteman R, Hamre LN, Helm A, Kelly R, Laanisto L, Lonati M, Moore JL, Morales M, Olsen SL, Pärtel M, Petry WK, Ramula S, Rasmussen PU, Enri SR, Roeder A, Roscher C, Saastamoinen M, Tack AJM, Töpper JP, Vose GE, Wandrag EM, Wingler A, Buckley YM. Global gene flow releases invasive plants from environmental constraints on genetic diversity. Proc Natl Acad Sci U S A. 2020 Feb 25;117(8):4218-4227. doi: 10.1073/pnas.1915848117. Epub 2020 Feb 7. PMID: 32034102; PMCID: PMC7049112.
- Lambert MR, Donihue CM. Urban biodiversity management using evolutionary tools. Nat Ecol Evol. 2020 Jul;4(7):903-910. doi: 10.1038/s41559-020-1193-7. Epub 2020 May 11. PMID: 32393868.
- DEMİR A. The impacts of climate change on genetic diversity. Biyolojik Çeşitlilik ve Koruma. 2021;14(3):511-518.
- Hao M, Zhang L, Ning S, Huang L, Yuan Z, Wu B. The resurgence of introgression breeding, as exemplified in wheat improvement. Front Plant Sci. 2020; 11:252.
- Davis AP, Chadburn H, Moat J, O'Sullivan R, Hargreaves S, Nic Lughadha E. High extinction risk for wild coffee species and implications for coffee sector sustainability. Sci Adv. 2019 Jan 16;5(1):eaav3473. doi: 10.1126/sciadv.aav3473. PMID: 30746478; PMCID: PMC6357749.
- Tao H. Transfer of Fusarium Head Blight Resistance to Hard Red Winter Wheat [Doctoral dissertation]. North Dakota State University; 2019.
- Dempewolf H, Baute G, Anderson J, Kilian B, Smith C, Guarino L. Past and future use of wild relatives in crop breeding. Crop Sci. 2017; 57(3):1070-1082.
- Behere GT, Tay WT, Russell DA, Heckel DG, Appleton BR, Kranthi KR, Batterham P. Mitochondrial DNA analysis of field populations of Helicoverpa armigera (Lepidoptera: Noctuidae) and of its relationship to H. zea. BMC Evol Biol. 2007; 7(1):1-10.
- Maxted N, Ford-Lloyd BV, Jury S, Kell S, Scholten M. Towards a definition of a crop wild relative. Biodivers Conserv. 2006; 15(8):2673-2685.
- Vincent H, Wiersema J, Kell S, Fielder H, Dobbie S, Castañeda-Álvarez NP. A prioritized crop wild relative inventory to help underpin global food security. Biol Conserv. 2013; 167:265-275.
- Dopman EB, Robbins PS, Seaman A. Components of reproductive isolation between North American pheromone strains of the European corn borer. Evolution. 2010 Apr 1;64(4):881-902. doi: 10.1111/j.1558-5646.2009.00883.x. Epub 2009 Nov 6. PMID: 19895559; PMCID: PMC2857697.
- Baack E, Melo MC, Rieseberg LH, Ortiz-Barrientos D. The origins of reproductive isolation in plants. New Phytol. 2015 Sep;207(4):968-84. doi: 10.1111/nph.13424. Epub 2015 May 5. PMID: 25944305.
- Smartt J. Gene pools in grain legumes. Econ Bot. 1984; 38(1):24-35.
- Bliss FA. Plant breeding, crop cultivars, and the nature of genetic variability. Intellectual Property Rights Associated with Plants. 1989; 52:69-89.
- Ogbonnaya FC, Abdalla O, Mujeeb-Kazi A, Kazi AG, Xu SS, Gosman N. Synthetic hexaploids: harnessing species of the primary gene pool for wheat improvement. Plant Breed Rev. 2013; 37:35-122.
- Pratt RC. Gene transfer between tepary and common beans.
- Upadhyaya HD, Sharma S, Dwivedi SL. Arachis. In: Wild Crop Relatives: Genomic and Breeding Resources. Springer, Berlin, Heidelberg; 2011; 1-19.
- Hammer K, Arrowsmith N, Gladis T. Agrobiodiversity with emphasis on plant genetic resources. Naturwissenschaften. 2003 Jun;90(6):241-50. doi: 10.1007/s00114-003-0433-4. Epub 2003 May 24. PMID: 12835833.
- Wilkes G, Williams JT. Current status of crop plant germplasm. Crit Rev Plant Sci. 1983; 1(2):133-181.
- Uzogara SG. The impact of genetic modification of human foods in the 21st century: a review. Biotechnol Adv. 2000 May;18(3):179-206. doi: 10.1016/s0734-9750(00)00033-1. PMID: 14538107.
- Snow AA, Andow DA, Gepts P, Hallerman EM, Power A, Tiedje JM, Wolfenbarger LL. Genetically engineered organisms and the environment: current status and recommendations. Ecol Appl. 2005; 15(2):377-404.
- Plucknett DL, Smith NJ. Gene banks and the world's food. Princeton University Press; 2014.
- Guo XD, Wang HF, Bao L, Wang TM, Bai WN, Ye JW, Ge JP. Evolutionary history of a widespread tree species Acer mono in East Asia. Ecol Evol. 2014 Nov;4(22):4332-45. doi: 10.1002/ece3.1278. Epub 2014 Oct 27. PMID: 25540694; PMCID: PMC4267871.
- Mamidi S, Rossi M, Moghaddam SM, Annam D, Lee R, Papa R, McClean PE. Demographic factors shaped diversity in the two gene pools of wild common bean Phaseolus vulgaris L. Heredity (Edinb). 2013 Mar;110(3):267-76. doi: 10.1038/hdy.2012.82. Epub 2012 Nov 21. PMID: 23169559; PMCID: PMC3668653.
- Larson G, Piperno DR, Allaby RG, Purugganan MD, Andersson L, Arroyo-Kalin M, Barton L, Climer Vigueira C, Denham T, Dobney K, Doust AN, Gepts P, Gilbert MT, Gremillion KJ, Lucas L, Lukens L, Marshall FB, Olsen KM, Pires JC, Richerson PJ, Rubio de Casas R, Sanjur OI, Thomas MG, Fuller DQ. Current perspectives and the future of domestication studies. Proc Natl Acad Sci U S A. 2014 Apr 29;111(17):6139-46. doi: 10.1073/pnas.1323964111. Epub 2014 Apr 22. PMID: 24757054; PMCID: PMC4035915.
- van Straalen NM, Timmermans MJ. Genetic variation in toxicant-stressed populations: an evaluation of the 'genetic erosion' hypothesis. Hum Ecol Risk Assess. 2002;8(5):983-1002.
- Van de Wouw M, Kik C, van Hintum T, van Treuren R, Visser B. Genetic erosion in crops: concept, research results, and challenges. Plant Genet Resour. 2010;8(1):1-15.
- Rogers DL. Genetic erosion no longer just an agricultural issue. Native Plants J. 2004; 5(2):112-122.
- Ahuja MR, Jain SM, editors. Genetic Diversity and Erosion in Plants. Springer; 2015.
- Rogers D, McGuire P. Genetic erosion: context is key. In: Ahuja MR, Jain SM, editors. Genetic Diversity and Erosion in Plants. Springer, Cham; 2015; 1-24.
- Jackson ND, Fahrig L. Relative effects of road mortality and decreased connectivity on population genetic diversity. Biol Conserv. 2011; 144(12):3143-3148.
- Begna T. Role and economic importance of crop genetic diversity in food security. Int J Agric Sci Food Technol. 2021; 7(1):164-169.
- Mew TW, Leung H, Savary S, Vera Cruz CM, Leach JE. Looking ahead in rice disease research and management. Crit Rev Plant Sci. 2004; 23(2):103-127.
- Bellon MR. Crop research to benefit poor farmers in marginal areas of the developing world: a review of technical challenges and tools. CAB Reviews: Perspectives in Agriculture, Veterinary Science, Nutrition and Natural Resources. 2006; 1(70):1-11.
- Koltunow AM, Bicknell RA, Chaudhury AM. Apomixis: Molecular Strategies for the Generation of Genetically Identical Seeds without Fertilization. Plant Physiol. 1995 Aug;108(4):1345-1352. doi: 10.1104/pp.108.4.1345. PMID: 12228546; PMCID: PMC157511.
- Swarup S, Cargill EJ, Crosby K, Flagel L, Kniskern J, Glenn KC. Genetic diversity is indispensable for plant breeding to improve crops. Crop Sci. 2021;61(2):839-852.
- Brozynska M, Furtado A, Henry RJ. Genomics of crop wild relatives: expanding the gene pool for crop improvement. Plant Biotechnol J. 2016 Apr;14(4):1070-85. doi: 10.1111/pbi.12454. Epub 2015 Aug 26. PMID: 26311018.
- Miller RE, Khoury CK. The gene pool concept applied to crop wild relatives: An evolutionary perspective. In: North American Crop Wild Relatives, Volume 1. Springer, Cham; 2018. 167-188.
- Anumalla M, Roychowdhury R, Geda CK, Mazid M, Rathoure AK. Utilization of plant genetic resources and diversity analysis tools for sustainable crop improvement with special emphasis on rice. Int J Adv Res. 2015; 3(3):1155-1175.
- Meffe GK. Conservation Genetics and the Management of Endangered Fishes. Fisheries (Bethesda). 1986 Jan;11(1):14-23. doi: 10.1577/1548-8446(1986)011<0014:CGATMO>2.0.CO;2. Epub 2011 Jan 9. PMID: 34170992; PMCID: PMC7164036.
- Jacobsen SE, Sørensen M, Pedersen SM, Weiner J. Feeding the world: genetically modified crops versus agricultural biodiversity. Agron Sustain Dev. 2013;33(4):651-662.
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.


 Save to Mendeley
Save to Mendeley
