International Journal of Aquaculture and Fishery Sciences
Histopathology of the Black Plaice Pseudopleuronectes Obscurus from a Marine Reference Area
National Scientific Center of Marine Biology, Far Eastern Branch, Russian Academy of Sciences, Vladivostok 690041, Russia
Author and article information
Cite this as
Syasina IG (2017) Histopathology of the Black Plaice Pseudopleuronectes Obscurus from a Marine Reference Area. Int J Aquac Fish Sci. 2017; 3(1): 12-17. Available from: 10.17352/2455-8400.000022
Copyright License
© 2017 Syasina IG. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.The Kievka Bay is one of the cleanest regions on the Far East coast of Russia. There are no anthropogenic impacts on the coastal area, and thus the influence of pollutants on the state of fish health is minimized. Histopathological examination of black plaice was performed to study the background level of morbidity and the occurrence of pathological alterations in tissues of fi shes from this ecologically clean region. Different pathological alterations were detected in fi shes sampled. The most surprising was a disease of black plaice, where necrotic changes were found in the gonads of 28% of the males, and the destructive changes were absent in females. In the gills, pathogenic microorganisms were detected, which induced strong hyperplasia of the respiratory epithelium, fusion of lamellas, and necrosis of pillar cells, in 28% of the individuals. Diseases of the liver were recorded for two fishes that comprised 8% of the total number, in one case – granulomatous inflammation. Besides that some fishes had a diffuse necrosis in the liver. In the kidneys of all sampled fish, parasites were present in the lumen of the renal duct, very likely a moderately pathogenic species, as the consequences of parasitism were insignificant and for a smaller part of fishes – moderate. Thus, different pathological alterations were found in all investigated organs, and, in most cases, it was possible to establish their occurrence with parasitism.
CRCN: Crustacyanin; H: Homogeneous Individuals; ST: Striped Translucent Shrimp
Introduction
Flatfishes are widely used as bioindicators of contamination of the marine environment [1-4] including moderate and heavily contaminated parts of Peter the Great Bay, Sea of Japan [5]. In our previous investigations we used endemic species – barfin plaice Liopsetta pinnifasciata and one of the most common species – black plaice Pseudopleuronectes obscurus [6]. The black plaice is a typical inhabitant of the upper subtidal. P. obscurus spends the greater part of its life in the near-shore zone, where various pollutants concentrate. Hence, it serves as an indicator species for the quality of the aquatic environment in Peter the Great Bay [7]. In terms of many parameters, the black plaice is more sensitive to pollution of marine environment than other species. Nevertheless, the occurrence of histopathological changes in this species from uncontaminated waters has not been studied. The aim of the present research was to evaluate, using biological, morphological, and histopathological indices, the state of the internal organs and tissues of fish from an uncontaminated reference marine area in Kievka Bay, Sea of Japan, in a case study of the plaice P. obscurus.
Materials and Methods
The investigation was conducted in a marine reference area in Kievka Bay, northwestern Sea of Japan (42°50’N, 133°37’-133°43’E). Our interest to this bay is explained by the fact that it is located in sparsely populated area adjoining the State Reserve of Laso, the forest reserve in Russian Far East. This presumes the absence of any important human impact on the environment in the chosen area of study. The subject of the investigation was a widespread species in the area of study, the black plaice Pseudopleuronectes obscurus (Herzenstein, 1890) (Syn. Pleuronectes obscurus). Fish with body lengths ranging from 18 to 37 cm were caught with a set net from 25 June to 1 July 2006 and 2007. The total number of fish studied was 28 in 2006 and 30 individuals in 2007. Before dissecting, skin and gill chamber were inspected for the presence of ectoparasites. Total length (TL), standard length (SL) and total body weight (W) were recorded for each animal. During the dissection the ovaries and liver were removed and weighed.
Condition factor (CF) was calculated according to Fulton’s formula: CF = (W × 100) / SL3, where W – total body weight, SL – standard length. Hepatosomatic index (HSI) was calculated according to: HSI (%) = (liver weight/total weight) × 100. Gonadosomatic index (GSI) was expressed as the percentage of body weight represented by the gonads: GSI (%) = (gonad weight/total weight) × 100. All values are expressed as the mean ± standard deviation (M ± SD). Data on CF, HSI and GSI are presented for fish collected in 2006, no significant changes of these parameters were found in 2007, so data are not shown.
Tissue specimens of liver, kidney, gills and gonad (0.8 × 0.8 см) were fixed with Bouin’s fluid for 48 h, dehydrated through a graded series of increasing concentrations of ethanol, cleared in xylene and embedded in paraffin. Sections (5-6 µm thick) were stained with eosin-hematoxilin by the standard method. The state of the tissues and organs were examined under a light microscope Olympus BX41 (Olympus, Japan) equipped with digital camera C5060-ADU (Olympus, Japan). Occurrence and classification of parasites in histological slides was established according to recommendations [8]. Liver tissues were analyzed for the presence of parasites, degenerative, inflammatory, necrotic, and preneoplastic changes, which were identified by generally accepted criteria [2,9].
Results
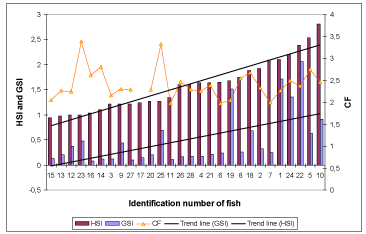
Condition factor of black plaice caught from 25 June to 1 July varied from 1.97 to 3.38, with a mean value of 2.44 (Figure 1). HSI ranged from 0.59 to 2.96, the mean in females 1.74 ± 0.66 (N = 9), and in males 1.48 ± 0.43 (N = 17). GSI ranged from 0.05 to 2.28. Low values of GSI are characteristic for fish in the “rest stage”, higher GSI up to 2.28 was found in two females in the “beginning of vitellogenesis stage”. In females, the mean GSI was 1.05 ± 0.61 (N = 9), and in males, it was 0.20 ± 0.13 (N = 17). Ectoparasites were not numerous. Copepods were observed on the skin and in the gill chamber of 3 fish, a 10.7% prevalence. For one black plaice, a skin growth on the fins was detected, presumably due to a trematode or nematode.
The state of the liver of P. obscurus caught in Kievka Bay corresponds to norm on the majority of indices. The hepatocytes were organized in hepatic tubules, the blood capillaries and sinusoids were not enlarged. The size of hepatocytes varied slightly, conditioned by the amount of stored nutrients. The nuclei and nucleolei were large enough, testifying to the high synthetic activity at this time of year. A few types of histopathological changes were registered in the liver of P. obscurus from Kievka Bay. The vacuolation of hepatocytes was observed in the liver of four fish for a prevalence of 14%. The edema of the liver of black plaice from Kievka Bay was not detected. Karyopyknosis was detected in 18% of the fish. Small areas of hepatocyte necrosis were detected in 14% of fish and inflammation in one fish (4%). Melanomacrophage centers (ММC) in the liver of plaice were few and small sized. The increase of their sizes was revealed in only one fish. Intensive granulomatous inflammation was characterized by formation of a granuloma and intensive leukocytic infiltration of enclosing tissues. This was well discernible in the background of hepatocytes with leucocytes migrating out the blood vessels to the damaged zone and surrounding the granuloma (Figure 2a,b).
A few types of histopathological alterations were present in the same individuals, for example, for fish № 15 – the increase of ММC, granulomatous inflammation, and necrosis of hepatocytes were detected, and for fish № 24 – necrosis, polymorphism of hepatocytes and their nuclei. Actually, only two fishes suffered disease of the liver.
In the gills of P. obscurus, a few types of histomorphological changes were observed, teratisms (frequency 7%), hyperplasia of the respiratory epithelium (18%) (Figure 3a), and parasitism (28%) (Figure 3b). In all cases the pathological changes were connected with presence of parasitic microorganisms that expressed a strongly pronounced tropism for the vascular system. The parasitophorous vacuoles microscopically were identical and were localized on lamella capillaries. The necrosis of pillar cells (confining capillary tubes) was localized close to vacuoles (Figure 3c). A second type of parasite, also at a very early stage of development, was localized inside of the cartilage of gill filaments. The gill parasites were not identified, but they have the definite tissue localization.
In the kidneys of all examined P. obscurus, parasites localized in the lumen of renal duct and/or glomerulus (prevalence 100%). The morphological structure of those parasites was representative of Myxozoa, “the cell in a cell”, the secondary cells (spores) localized inside primary cells was observed (Figure 4a). The damage induced by parasites was mostly minor although moderate in several individuals; single necrotic cells were found in the renal duct and sometimes they filled the whole renal duct. Multifocal distributed granulomas were noted in the kidney stroma. The granulomas consisted of densely disposed macrophages and epithelioidal cells, with necrosis in the central part of the granulomas (Figure 4b).
In the gonads of two females, the process of vitellogenesis had been started, while the gonads of all other females and all males were in the resting stage. Significant pathological alterations were detected in the gonads of black plaice from Kievka Bay. Necrotic changes were detected in 28% of males. In pathologically changed tubules, very large, presumably spermatogenic cells, were present that broke apart when they reached this size (Figure 5a). The cellular debris filled the seminiferous tubules, yet were not broken down with nuclei floating in the unstructured mass. In injured testis the zones of hemorrhages and occasional severe inflammation were noted (Figure 5b). The damage to the seminiferous tubules started at the surface of the testis (Figure 5c), and spread from the surface deep into the gonad (Figure 5d), thus the gonad was reshaped by superficial constriction.
Discussion
Biomonitoring of flatfish health has been conducted by a scientific team from the A.V. Zhirmunsky Institute of Marine Biology, now National Scientific Center of Marine Biology, in the coastal zone of Peter the Great Bay since 1995. Flatfish from the southwest part of Peter the Great Bay and Amursky Bay have been investigated in detail, with histopathological study of the internal organs and vitellogenin measurements sometimes performed [6,7,10]. Histopathological examination of fish from Kievka Bay was achieved for the first time. Kievka Bay is one of the cleanest regions on the Far East coast of Russia. There are no anthropogenous inputs, and thus the influence of pollutants on the state of fish health is minimized. The present study determined condition factor according to Fulton, hepato- and gonadosomatic indices on one stage of the reproductive cycle. Because the sampling dates for black plaice collections have differed between this and other regions, it is not possible to compare the data from this study to other investigations. This information may be beneficial later as more analyses of fishes from contaminated regions takes place.
Different pathological alterations were detected in the internal organs of black plaice P. obscurus from uncontaminated reference area in Kievka Bay. The most surprising fact is the disease of the black plaice, where necrotic changes in male gonads were marked, while the destructive changes were absent in females. The disease was noted in 2006 and 2007, with the prevalence about identical at 28%. On the diseases of fish from this water area is not known, but there is evidence that in the sea urchin Strongylocentrotus intermedius found spawning failure [11].
The pathology of the gonads has been frequently recorded in fish inhabiting the contaminated areas. Cases of large increase or decrease in gonad volume (sometimes their full absence), asymmetry of the paired gonads (at worst – the development only of one), and asynchronous maturation have been described. In male gonads, connective tissue constrictions can be formed and, as a consequence, – the formation of the twisted gonads, tucks, and segmentation. In some cases, dense connective tissue appeared that resulted in the occlusion of the semen duct and degeneration of gonads [12]. Also, it is possible to observe the gelatinous condition of the female gonads and the unusual pigmentation of the gonads [13]. None of these pathologies of the gonads were detected in P. obscurus from Kievka Bay.
In previous studies, the pathological changes of the gonads in flatfish from Peter the Great Bay have been recorded only in two cases. In female barfin plaice Liopsetta pinnifasciata, living in the Amursky Bay, one of the most polluted areas in Peter the Great Bay of the Sea of Japan, abnormal oogenesis was described, characterized by mass atresia of ovarian follicles in the spawning period [14]. In male L. pinnifasciata from the same region the gonad pathology was exhibited in the appearance in the testis of melanomacrophages centers and pigmented granules in Sertoli cells. Such phenomena were seen in 20% of males and suggest the destruction of cells, or the presence of a pathogen. For 4% of the fish in the study that measured 20 to 25 cm, a length where individuals should be puberal, the phenomena of a hypogonadism (depression of the testes function, reduction, or their full absence) [15] was noted. The authors remarked that the degree of gonad differentiation was very low: in the central part of the testes the seminiferous tubules with lumen were present, close to the peripherals – the lumen of tubules were narrowed down and then were completely closed. The proliferation of Sertoli cells was noted along the walls of tubules or partially occluding the lumen. Along with the increase in the number of Sertoli cells (hyperplasia), their volume increase (hypertrophy) was also marked. The nuclei of Sertoli cells in abnormal testis were augmented by 25% as contrasted to those in a normal gonad. Spermatogenesis was lacking with only single spermatogonia detected. This type of testis pathology very seldom occurs in fish. Isolated instances are described and identified by the terms hypogonadism [15] and testis dysgenesis [16]. These types of testis pathology are not characteristic for P. obscurus from Kievka Bay. Thus, the testis disease in black plaice from Kievka Bay was detected for the first time. The necrotic changes were observed only in the gonads of males, not females. Comparative analysis of histopathological observations of the testis of black plaice from Kievka Bay shows that a similar disease was not seen in black plaice or in other flatfish species from Peter the Great Bay.
Pathology of the gonad of fish from contaminated areas is characterized by specific types of changes, in particular in phenotypic male flounder Platichthys flesus from the Mersey estuary (UK) impacted by endocrine disrupting chemicals, uni- and bilateral intersex gonads with abnormal ovarian follicular components in the testis were observed [17]. Reduced testicular growth and sperm production was observed in male fish under experimental exposure to contaminants, for example in male rainbow trout Oncorhynchus mykiss exposed to four different alkylphenolic chemicals [18]. Very specific pathology of the testes or sperm ducts was observed in spotted gar Lepisosteus oculatus from Bayou Trepagnier, Louisiana, USA, a petroleum-contaminated site, multilocular fluid-filled cysts [19].
The etiology of disease is not established yet. Examples of bacterial or viral infections, where the fish testis only would be affected have not been reported. There are other groups of pathogenic agents that do affect the testis. Among myxosporidians there are species that infect the gonads of males. Examples include Sphaerospora testicularis [20] and Henneguya testicularis [21]. The first of these parasites, S. testicularis, induces the destruction of seminiferous tubules, the decrease in a number or full elimination of mature spermatozoa. The pathogenic effect of the second species, H. testicularis, was not indicated. Additional studies are required for detection of a possible pathogenic agent of the testis necrosis noted in this study.
There were granulomas in the kidneys and, in one case, the liver of black plaice from Kievka Bay. The formation of multiple granulomas is denoted by the term a granulomatosis. Granulomatosis is a nonspecific type of an inflammation that is induced in fishes by a variety of the different agents. A classic example is the inflammatory response in fishes, as well as in more highly organized vertebral animals, to bacterial infections by the genus Mycobacterium [22, 23]. Granulomatosis is induced also by other organisms, in particular some endocellular bacteria, Dermocystidium-like parasites [24] and Ichthyophonus hoferi [25]. The formation of a granuloma contributes to the control of pathogens, which cannot be eliminated by phagocytosis or digested by macrophages. The granulomas in fish represent the compact, organized accumulation of mature monocytic phagocytes (macrophages and/or epithelioidal cells), although the cellular composition of granulomas can vary. In a black plaice, the forming of granulomas was tracked by a powerful infiltration of enclosing tissues by leucocytes that suggests a strong immune response. Almost all detected granulomas had a necrotic center.
Conclusion
Different pathological alterations were found in organs of black plaice from Kievka Bay, northwestern Sea of Japan. In most cases it was possible to associate these alterations with parasitism. The principal findings are necrosis in the testis, hyperplasia of the respiratory epithelium, fusion of gill lamellas, necrosis of pillar cells in gills, and granulomatous inflammation in the kidney and liver. The spectrum and occurrence of histopathological changes were lower than in flatfish from contaminated parts of Peter the Great Bay, Sea of Japan. Several of the observed abnormalities in this study were found in flatfish from Russian part of the Sea of Japan for the first time.
I would like to thank Dr. R.G. Getchell for kindly correcting the English.
- Vethaak AD, Bucke D, Lang T, Wester PW, Jol J, et al. (1992) Fish disease monitoring along a pollution transect: a case study using dab (Limanda limanda) in the German Bight. Mar Ecol Progess Ser. 91: 173–192. Link: https://goo.gl/S341MF
- Vethaak AD, Wester PW (1996) Diseases of flounder Platichthys flesus in Dutch coastal and estuarine waters, with particular reference to environmental stress factors. II. Liver histopathology. Dis Aquat Organ. 26: 99–116. Link: https://goo.gl/FM0F3c
- Leonardi M, Vera J, Tarifeño E (2009) Diseases of the Chilean flounder Paralichthys adspersus (Steindachner, 1867) as biomarkers of marine coastal pollution near the Itata River (Chile). Part I: In Situ macroscopic lesions. Arch Environ Contam Toxicol 56: 536–545. Link: https://goo.gl/B4H6NI
- Oliva M, Vicente-Martorell JJ, Galindo-Riano MD, Perales JA (2013) Histopathological alterations in Senegal sole, Solea senegalensis, from a polluted Huelva estuary (SW, Spain). Fish Physiol Biochem 39: 523–545. Link: https://goo.gl/7NAtk8
- Vaschenko MA, Syasina IG, Durkina VB, Zhadan PM (2003) Contamination of sediments and histological alterations in barfin plaice Pleuronectes pinnifasciatus from Amursky Bay (Peter the Great Bay, East Sea/Sea of Japan). Ocean Polar Res 25: 31–40. Link: https://goo.gl/D3VVfQ
- Syasina IG, Arbuzova LL, Zhad’ko EA, Sokolovskii AS (2000) Histomorphological alterations in organs of the plaice Pleuronectes obscurus from a polluted part of Peter the Great Bay, Sea of Japan. Russ J Mar Biol 26: 276–282. Link: https://goo.gl/kny4Lz
- Syasina IG, Sokolovskii AS (2001) Assessment of the state of flounders from Sivuch’ya Bight (Peter the Great Bay, Sea of Japan) based on histopathological parameters. Russ J Mar Biol 27: 80–86. Link: https://goo.gl/O2xaOo
- Bruno DW, Nowak B, Elliott DG (2006) Guide to the identification of fish protozoan and metazoan parasites in stained tissue sections. Dis Aquat Organ 70: 1-36. Link: https://goo.gl/0aQmLQ
- Hinton DE, Baumann PC, Gardner GR, Hawkins WE, Hendricks JD, et al. (1992) Histopathologic biomarkers. In Biomarkers. Biochemical, physiological and histological markers of anthropogenic stress, Edited by Huggett RJ, Kimerle RA, Mehrle PM, Bergman HL, Boca Raton, Lewis Publishers, FL, 155–209. Link: https://goo.gl/4g7g0n
- Shved N, Kumeiko V, Syasina I (2011) Enzyme-linked immunosorbent assay (ELISA) measurement of vitellogenin in plasma and liver histopathology in barfin plaice Liopsetta pinnifasciata from Amursky Bay, Sea of Japan. Fish Physiol Biochem 37: 781–799. Link: https://goo.gl/TKHhNm
- Zhadan PM, Vaschenko MA, Almyashova TN (2015) Spawning failure in the sea urchin Strongylocentrotus intermedius in the northwestern Sea of Japan: Potential environmental causes. J Exp Mar Bio Ecol 465: 11–23. Link: https://goo.gl/1GVVyZ
- Blazer VS (2002) Histopathological assessment of gonadal tissue in wild fishes. Fish Physiol Biochem 26: 85–101. Link: https://goo.gl/k91Fnz
- Kashulin N, Lukin A, Amundsen PA (1999) Fish of Subarctic freshwater systems as bioindicators of industrial pollution. Apatity.
- Durkina VB, Cherkasova I V. (2007) Abnormal oogenesis in barfin plaice Pleuronectes pinnifasciatus from the Amursky Bay of the Sea of Japan. Biol Bull 34: 492–497. Link: https://goo.gl/X6ikz8
- Syasina I, Vusotskaya V, Shved N (2006) Histopathological changes in the liver and gonads of barfin plaice Pleuronectes pinnifasciatus from the Amursky Bay. In Proceeding of International scientific-practical conference. Ecological problems related to use of coastal marine waters, 26-28th October 2006, Vladivostok, 188–191.
- Hecker M, Murphy MB, Coady KK, Villeneuve DL, JonesPD, et al. (2006) Terminology of gonadal anomalies in fish and amphibians resulting from chemical exposures. Rev Environ Contam Toxicol 187: 103–131. Link: https://goo.gl/9wmiPW
- Simpson MG, Walker P, Helm A, Leah R (2002) Histopathological observations on liver, kidney and gonad of plaice (Pleuronectes platessa) taken from the Mersey estuary. Mar Environ Res 54: 543–546.Link: https://goo.gl/ehffsy
- Jobling S, Sheahan D, Osborne JA, Matthiessen P, Sumpter JP (1996) Inhibition of testicular growth in rainbow trout ( Oncorhynchus mykiss ) exposed to estrogenic alkylphenolic chemicals. Environ Toxicol Chem 15: 194–202. Link: https://goo.gl/XUFWai
- Thiyagarajah A, Anderson MB, Hartley WR (2000) Gonadal cysts in spotted gar (Lepisosteus oculatus) from Bayou Trepagnier, Louisiana, USA. Mar Environ Res 50: 279–282. Link: https://goo.gl/4vGUPy
- Sitja-Bobadilla A, Alvarez-Pellitero P (1990) Sphaerospora testicularis sp. nov.(Myxosporea: Sphaerosporidae) in wild and cultured sea bass, Dicentrarchus labrax (L.), from the Spanish Mediterranean area. J Fish Dis 13: 193–203. Link: https://goo.gl/RHVPjY
- Azevedo C, Corral L, Matos E (1997) Light and ultrastructural data on Henneguya testicularis n. sp. (Myxozoa, Myxobolidae), a parasite from the testis of the Amazonian fish Moenkhausia oligolepis. Syst Parasitol 37: 111–114. Link: https://goo.gl/RiOBJs
- Chan K, Knaak T, Satkamp L, Humbert O, Falkow S, et al. (2002) Complex pattern of Mycobacterium marinum gene expression during long-term granulomatous infection. Proc Natl Acad Sci 99: 3920–3925. Link: https://goo.gl/jBmk1R
- Swaim LE, Connolly LE, Volkman HE, Humbert O, Born DE, et al. (2006) Mycobacterium marinum infection of adult zebrafish causes caseating granulomatous tuberculosis and is moderated by adaptive immunity. Infect Immun 74: 6108–6117. Link: https://goo.gl/q1XboF
- Landsberg J, Paperna I (1992) Systemic granuloma in goldfish caused by a Dermocystidium -like aetiological agent. Dis Aquat Organ 13:75–78. Link: https://goo.gl/7bnkKg
- Gavryuseva TV (2007) First report of Ichthyophonus hoferi infection in young coho salmon Oncorhynchus kisutch (Walbaum) at a fish hatchery in Kamchatka. Russ J Mar Biol 33:43–48. Link: https://goo.gl/dJm9Tz
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.






 Save to Mendeley
Save to Mendeley
