International Journal of Aquaculture and Fishery Sciences
Enhancement of aquaculture performance of cobia, Rachycentron canadum (Linnaeus 1766): A review
Department of Biology, Umaru Musa Ya’radua University, P.M.B. 2218, Katsina State, Nigeria
Author and article information
Cite this as
Ademola BT (2024) Enhancement of aquaculture performance of cobia, Rachycentron canadum (Linnaeus 1766): A review. Int J Aquac Fish Sci. 2024; 10(1): 1-8. Available from: 10.17352/2455-8400.000090
Copyright License
© 2024 Ademola BT. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Cobia, Rachycentron canadum, is a marine fish with recognized potential for aquaculture due to its fast growth rate, high fecundity, disease resistance, and quality flesh. However, much progress has been made to overcome problems associated with aspects of cobia production, including broodstock management, fingerling production, larval-rearing, grow-out, and disease management, though, an effort is needed to ensure its sustainability. Broodstock conditioning protocols for continuous spawning as well as induced spawning via hormonal injection resulted in all-year-round seed production in cobia, but detailed information on the cost-effective dosages of various spawning hormones for maximum fecundity and larval survival needs to be investigated. Selective breeding, the application of genetics in trait selection, and the use of molecular markers could produce more viable strains to mitigate high mortality, particularly in colder climates. In addition, considering the fact cobia are carnivorous fish with a high protein requirement, it will be necessary to investigate fishmeal alternatives to reduce feeding costs. Low salinity pond culture may become a reality in the future if the cobia metabolism-physiology of nutrition at various levels of salinity is well understood. The present paper provides information on some of the options for optimizing the aquaculture performance of Rachycentron canadum.
Among emerging species with potential for commercial production is cobia, Rachycentron canadum. Cobia is the only species of the family Rachycentridae that is widely distributed throughout the subtropical, tropical, and temperate waters, except for the eastern Pacific region [1,2]. They are migratory and pelagic fish, they move to deeper waters in the fall and winter [2]. Cobia is a non-target species in fisheries as most of their landings were from recreational fishing. However, countries, where cobia capture is high, exceeding 1000 metric tons per annum, are Pakistan, Iran, Malaysia, and Brazil [3] (Figure 1). Meanwhile, cobia constituted 0.1% of the landings between 2007 and 2010 on the Karnakata coast, of India [4].
Cobia exhibits a benthopelagic lifestyle and is a non-selective feeder that searches for prey from the benthic zone and open water [4-7]. Most of the studies on the feeding habits of cobia focus on juveniles and adults, while the natural food of young cobia in the wild has not been reported, but it was believed that they feed on copepods [8]. Naturally, cobia exhibit extended batch spawning, and peak spawning seasons vary with geographical locations. Cobia has biological characteristics which make it suitable for mariculture, showing fast growth up to 6 kg in one year in captivity [9], a high growth rate (K) = 2.6 (yr-1) in the wild [10] and high fecundity up to 2.88 × 106 [11]. The value of feed conversion ratio (FCR), a measure of the ratio between feed input and output of 1.05 has been reported for cobia in the juvenile stage [12] and FCR of 2.0 in adults [13]. In addition, cobia are resistant to disease and possess excellent flesh that commands a high market price [14].
The first attempt to culture cobia using naturally spawned eggs collected from the wild was carried out by Hassler and Rainville [15]. Its performance in the 131-day rearing trial from hatching rate, growth rate, readiness to accept supplemented feed, and hardiness, led to the conclusion that cobia has a potential for culture.
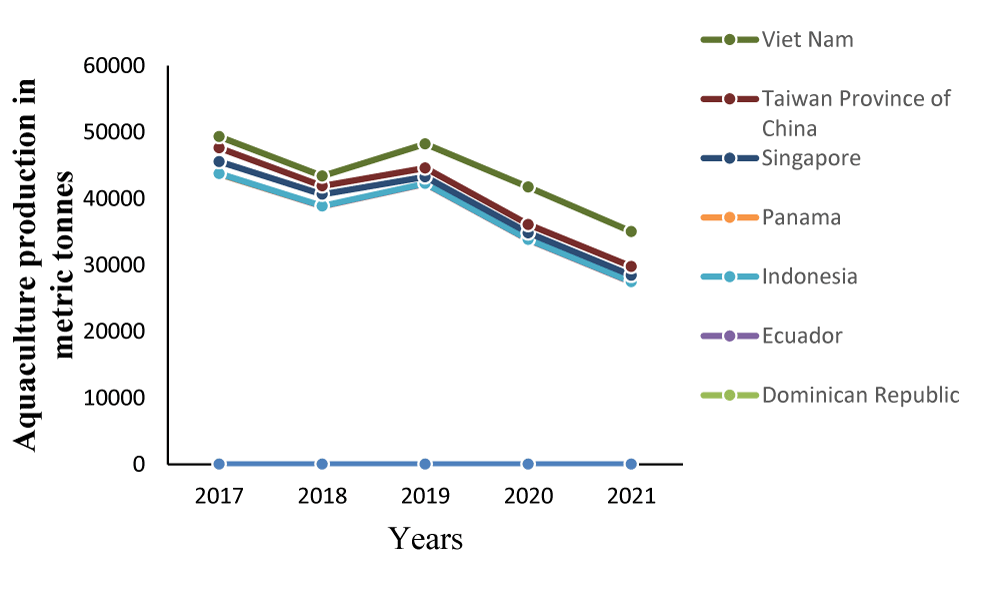
The cobia aquaculture expanded between 1995 to 2012 [16]. Currently, aquaculture production has leveled up in some countries and diminishing in others [17] (Figure 2). Year-to-year variation in cobia production in different countries was described as mixed, as the trends are generally fading due to production bottlenecks [18]. Nhu, et al. [19] reviewed various aspects of cobia production technology adopted in Vietnam. Broodstock conditioning and induced spawning were employed for larvae production in the country. Both intensive rearing in recirculating aquaculture systems and semi-intensive in the outdoor pond have been developed for larviculture of cobia in Taiwan [8].
Among the identified problems mitigating the commercial production of cobia include: a lack of consistent fingerlings production, larvae survival, mortalities during winter in some regions, and disease outbreaks. In addition, the utilization of alternative proteins, especially those of plant origin by cobia in the adult stage remains an issue. This review, therefore, elucidates some of these problems and suggests a way forward to enhance the performance of cobia for increasing commercial production.
Broodstock selection and improvements
For breeding, cobia broodstocks were mostly obtained from the wild [20]. Since fast growth is one of the desired traits in culture industries, investigating and comparing the natural growth of the wild population plays a crucial role in the selection of broodstock. Appropriate broodstock development and management for the production of a high quantity of fertilized eggs requires the knowledge of conditions for gonad maturation and spawn induction. Population characteristics of cobia in different geographical locations have been reported to exhibit differences in growth rate, sex ratio, and size at first maturation, and lifespan (Table 1). The growth rate (K) reported for cobia in northern and eastern Australia was 0.63 yr-1 [25]. Smith [7] found that the K value for cobia in North Carolina waters was 0.37. Therefore, the development of broodstock from the best population is indispensable.
Genetic assessment of cobia for stock identification has been documented for some geographical areas while the use of genetic markers for trait selection and broodstock development has not been reported. For example, Gold, et al. [26] utilized both nuclear-encoded microsatellite genotypes and mtDNA sequences to establish genetic differences in cobia populations from the Northern Gulf of Mexico, U.S. Western Atlantic, and Taiwan, Southeast Asia. Of the 28 nuclear-encoded microsatellites used, the result obtained showed that cobia from the Northern Gulf of Mexico and U.S. Western Atlantic waters were homogeneous for alleles and genotype at 27 nuclear-encoded microsatellites, as the exact tests for alleles and genotypes were not significantly different, while Taiwan population differed considerably. The same pattern was obtained in mtDNA sequences used. The author suggested that transferring broodstock from Taiwan to U.S. water should be done with caution. A similar scenario of a homogenous population of cobia was reported in the southeastern U.S. Atlantic Ocean by Darden, et al. [27], where tagging, genotyping, and parentage analysis, supported a single cobia population in the area despite the discreetness of a local estuarine population. In another study, nine of the most polymorphic nuclear-encoded microsatellites amongst those developed by Pruett, et al. [28] were applied to study the genetic diversity and population structure of the cobia population in the Gulf of Thailand and the Andaman Sea [29]. Population homogeneity of cobia from the sample area was observed and this was adduced to the migration and mixing of resident and transient groups, which resulted in homogeneity in allele frequencies on a larger geographic scale. The author opined that this scenario may extend to about eight countries: Myanmar, India, Thailand, Malaysia, Indonesia, Singapore, Cambodia, and Vietnam. Thus, management of the cobia population and genetic conservation in the area could be considered at the regional level and the transfer of broodstock within this region may not be discouraged. An attempt was made to culture cobia in cages in Malaysia using broodstock obtained from Taiwan, although it resulted in total mortality [30]. This may be due to transportation stress.
The application of genetics for trait selection and improvement like parentage assignment and estimation of heritability for an important trait has not been used for cobia. Desirable traits like higher feed conversion efficiency, plant protein utilization efficiency, fast growth, disease resistance, and cold tolerance could be selected in future broodstock development for cobia. Genetics have been applied for trait selection in Nile tilapia for improved growth in brackish water systems in Vietnam [31]. Three strains of tilapia namely GIFT (Genetically Improved Farmed Tilapia), Taiwan strains, and NOVIT4 (GIFT-derived) strains were selected for the best-performing individuals in terms of growth and then crossed. Free breeding between and within the groups was conducted over a few generations and the genetic analyses of the resultant population showed improvement in harvest weight. The development of cold tolerance stock using such protocol will be of great importance in cobia production in many regions where cobia growth is being limited by winter. It has been reported that reduced temperature causes severe growth inhibition and mortality in all life stages of cobia [8]. Selective breeding can also enhance the cold tolerance of cobia using specimens that survived the winter to produce next-generation offspring. These can be used in the selective breeding program using microsatellite markers linked to traits of interest, or cold tolerance in this case. Since the cold tolerance trait has been identified as an additive inheritable trait [32].
Production of cobia seed
The spawning period of cobia in the wild (Table 1), indicated that cobia exhibit extended batch spawning and peak spawning seasons vary with geographical locations. A spawning interval of 7.6 days was reported for cobia in northeastern Australia [11], 5 days in the southeastern United States and the north-central Gulf of Mexico, and 9 – 12 days in the western Gulf of Mexico [23]. The variations in spawning frequency in the wild and the peak of the spawning season may be controlled by temperature, distances covered to spawning grounds, and habitat productivity. Understanding the influence of habitat parameters, most importantly temperature, on spawning frequencies of cobia could be utilized in optimizing their spawning in captivity. Induced spawning was reported by Arnold, et al. [33] using a photoperiod of 13:11 and 14:10 light to dark (L: D) while the temperature ranged from 24.5 to 28.5 C. The study observed increased activity in cobia as the water temperature approached 26 – 27 C and subsequently spawning took place in two months, once each month, resulting in a hatch rate of up to 83 %. Kaiser and Holt [34] reported monthly continuous captivity spawning of cobia for up to 9 months with a photoperiod ranging from 10:14 to 14:10 (L:D) and water temperature ranging from 20 °C – 26 °C. Natural spawning of cobia for a year-round via environmental manipulation was reported by Stieglitz, et al. [35]. Broodstock was held in 80 m3 Recirculating Aquaculture Systems (RAS) and the temperature was maintained at 27 °C – 29 °C for 12 months, during which cobia spawn continuously. In addition, this work showed a high association between the spawning event and environmental temperature since no spawning event took place during winter and fall in an uncontrolled broodstock tank, whereas spawning activities were recorded every 14.4 days in the on-season period and every 9.2 days in the off-season period. The factor of photoperiod appeared not to interfere in any way with the spawning event thus, cobia spawning was found to be attuned with environmental temperature and not photoperiod. The interval between the spawning events was still higher compared to what had been reported in the wild.
Induced reproduction via hormonal administration has been well-reported. A single dose of human chorionic gonadotropin (hCG) at 500 UI for females and 250 UI for males induced spawning in cobia [14]. This produced approximately 2.1 million eggs from a single female with up to 90 % fertilization and about 80 % hatching. Franks, et al. [36] utilized hCG at the rate of 275 IU.g-1 for female cobia, and recorded a fecundity of 3.1 million eggs for the two females combined, although fertilization and hatching rates were not recorded. Gopakumar, et al. [14] observed spawning at about 39 hours after the administration of hormone while in Franks, et al. [36] it was 42 hours. Table 2 compares the hatching rates of cobia under natural and hormonal-induced spawning. Nhu [39] administered luteinizing hormone (LH–RHa) at a dose of 20 μg.kg−1 female and 10 μg.kg−1 male to induce spawning in cobia and the result showed a similar fecundity and fertilization rate comparable with the natural method. An appropriate dosage of the minimum-inducing hormone for ovulation has been noted to produce a maximum hatch rate [40]. Hence, further research may be needed to provide information on the lowest dose required to induce ovulation in cobia for maximum fecundity and hatching. Through the combination of hormonal and environmental induced spawning, a consistent production of seed could be achieved in other to establish hatcheries for an all-year-round supply of fingerlings.
Rearing and environment
Cobia adapts to rearing in ponds [41], offshore and nearshore cages [34,42,43], raceway systems [44] and recirculating aquaculture systems [45]. This presents an additional opportunity for farmers to make a choice. Cobia mariculture in Taiwan, China, and Vietnam where production was high showed that cage culture (floating or submerged) was mostly used [46]. Fishes were held in open water, where water temperature, salinity, and other parameters were not controlled but varied with season, weather, and the time of day. Natural phenomena like typhoons and winter colds cause mortality. Salinity fluctuations remain within the tolerance range for cobia. Mass mortality has been recorded when the temperature falls below 16 C in winter [8]. This has been limiting cobia production. The development of a cold-tolerant strain of cobia and the adoption of an intensive system like RAS where water temperature could be controlled is essential.
The suitability of Recirculating Aquaculture Systems (RAS) for cobia production has been established. Hitzfelder, et al. [45] reported a negative correlation between stocking density and the growth of cobia larvae in an intensive system. While no significant differences in survival rate and length increase for cobia larvae reared at the low-density of 0.5, 1.1, and 2.0 larvae per liter of water. In addition, survival was significantly higher in the low-density. To achieve a cost-effective RAS for cobia, optimal rearing density for fillet output needs to be well understood and possibly in low salinity.
Weirich, et al. [47] compared biomass production and body composition of juvenile cobia reared in RAS using natural saltwater. The study suggested that cobia tolerates rearing in RAS up to about 2 kg body weight at densities ≤ 30 kg.m-3, as the survival rate was high (≥ 96 %) and feed conversion efficiencies ranged from 65 % to 85.7 %. The RAS for cobia production could alleviate some of the problems associated with open sea culture in a place like the U.S. where effluent discharge regulations and restrictions on the use of coastal and offshore environments are strict [48].
Nutrition
Cobia, in their natural habitat, exhibits opportunistic carnivorous feeding, and varieties of prey items have been recorded in their stomachs in different studies [4,6,49,50]. Ganga, et al. [10] recorded five species of mollusk, five species of crustacea, and nineteen species of teleost in the diet of cobia in the south-eastern Arabian Sea, west coast of India. Fish had the highest percentage dominance of 90 %. Arendt, et al. [51] recorded one species of bivalve, one species of hydroid, six species of crustacean, one elasmobranch, and 16 species of teleost in the stomach content of cobia in the lower Chesapeake Bay, US. While this information can be potentially utilized for cobia broodstock feeding, currently, there is no commercially available cobia broodstock feed. Nguyen, et al. [52] compared the use of Raw Fish (RF) and three formulated diets of similar proximate composition with varying n-3 unsaturated fatty acids on the performance of cobia broodstock. The results showed no significant differences in the spawning success observed among dietary groups. The fatty acid content of spawned eggs was found to vary proportionally to that of the diet. A Higher Arachidonic Acid (ARA) content in eggs was determined to contribute to lower fertilization. The study also showed that cobia broodstock requirements of dietary n-3 Polyunsaturated Fatty Acids (PUFA) should be higher than 1.86 % diet dry weight and that high dietary levels of ARA above 0.42 % dry weight of the diet may affect cobia fertilization success negatively. Egg and larvae quality have been identified to be impacted by the broodstock diet. In Turkey, Sea bass broodstock feeding protocol utilized pelleted feed containing crude protein > 45 %, lipids > 16 %, n3-HUFA > 40 mg.m-1 dry weight of diet, DHA > 26 mg.g-1, EPA > 13 mg.g-1 and carbohydrate < 15 % in the final stage of the egg development, as this was found to be optimal for seed production and survival [53]. Therefore, training cobia broodstock to accept pellet feed, containing adequate nutritional components will optimize egg quality for improving the larval survival.
In the cobia life history, morphological changes in the digestive tract took place in the first 1–4 days post-hatching (DPH ) when the larvae were about 3.6 – 4.4 mm and beginning to absorb the yolk which became exhausted by 5 DPH [44]. In intensive systems of cobia rearing, larvae are often fed with rotifers as a first food followed by Artemia. Faulk and Holt (2003) [54] subjected cobia larvae to Artemia, enriched rotifers, and microparticulate diet in different combinations and varied starting periods after hatching. There were no significant differences in the growth of cobia-fed rotifers at the end of the 13-day trial. However, the administration of microparticulate feed for cobia after one day as well as the use only Artemia resulted in poor growth and total mortality. Therefore, feeding with rotifers for a few days before Artemia or a microparticulate diet was found to be essential in cobia seed production.
Under rearing conditions, cobia larvae start to feed on rotifers at 3 DPH and on newly hatched Artemia nauplii by 5 DPH. The cobia larval development on copepods as first feed was faster than rotifers and Artesia. Cobia larvae utilized copepods as food in a natural environment. This is why cobia larvae culture in enriched outdoor ponds was preferred before transferring to sea cages or other rearing conditions, although mortality of larvae was often higher [8]. In the larval fish-rearing tank, the movement of the live food organisms can stimulate fish-feeding behavior [55] and copepods perform better in attracting cobia larvae. It has also been proven that copepods have significantly higher concentrations of highly unsaturated fatty acids (HUFA) compared to rotifers and Artemia [56] in addition to digestive enzymes present in live food [57].
Cobia larvae metamorphose from cutaneous respiration to gill respiration at 11-15 DPH [58]. This metamorphosis was accompanied by morphological changes as the mouth becomes functional and the eyes develop pigmentation [59]. This stage is crucial in the process of weaning onto the commercial diet. Faulk, et al. [44] utilized rotifers 3-7 dph followed by Artemia 7-10 dph before weaning onto formulated feeds and this resulted in low larval survival of 13.2 ± 3.2 % at day 29 dph. Perhaps using rotifers for an extended period before Artemia will be required for better survival of cobia larvae. Therefore, intensive larval rearing using rotifers as a first feed before feeding with newly hatched Artemia is not the only essential aspect of achieving better growth and survival of larval, understanding the duration of feed with these live organisms is also crucial.
The juvenile stage starts from about 20 DPH when larvae reach 16.4 mm standard length and the stomach is fully differentiated [41]. This observation supported Ditty and Shaw [59] who reported a full complement of rays in all fins of cobia around 20 mm standard length, at the beginning of the transition to the juvenile stage. At this stage, sufficient protein inclusion in diets for growth is vital as carnivorous fish species would require high protein feed in captivity. The protein and lipid requirement of juvenile cobia as determined by Chou, et al. [9] was about 45 % and 5.76 % respectively. Most commercial cobia feed in Taiwan contains a crude protein concentration of about 45.3 %. A high lipid level of 16 % was tested by Zink, et al. [60] and this did not contribute to growth improvement.
Protein is the most important and expensive component of the fish diet. Its quality, which is dependent on the source, determines the suitability of such feed in aquaculture. Fishmeal is a superior protein source for fish since it has a well-balanced amino acid profile with no anti-nutritional factors that are present in many fish meal alternatives. However, as demand for fish increases and overfishing becomes more apparent, which results in a high price and limited supply, finding alternative protein sources, rather than using fish to feed fish becomes necessary. One of the major problems in the cobia farming industry is the high requirement of protein and the resultant increase in production cost. Hence, there is a need for alternative protein sources for cobia. Generally, several alternate protein sources have been identified for aquaculture feed and some of these protein sources have been tested in cobia and the results showed that cobia can utilize a wide range of protein sources of animal and plant origin without affecting its nutritional value.
In a study by Lunger, et al. [61] which evaluated the utilization of yeast-based protein as a replacement for fish meal in diets for juvenile cobia, it was concluded that replacement of up to 25 % of dietary protein can be provided by the yeast-based protein without any adverse effect on cobia. The performance of juvenile cobia on two common commercial diets and a plant-based-protein-based diet resulted in no significant differences in weight gain, feed conversion ratio, protein gain, and plasma amino acid in cobia fed [62]. However, cobia fed with a commercial diet had more lipid deposits than those fed with a plant-based diet. Watson, et al. [63] studied the growth performance and the lipid contents of juvenile R canadum. In the study, cobia were fed with taurine supplemented plant protein diet with fish oil replaced with the Thraustochytrid Meal (TM) Plus Soybean Oil (SOY) in one diet (TM + SOY), fish oil replaced with a canola oil supplemented with essential fatty acids (EFA) {Docosaexanoid Acid (DHA) and Arachidonic Acid (ARA)} in the second diet (CAN + EFA), while the third diet was a plant-based protein cobia feed containing fish oil as the lipid source as the control diet (ARS). Both (TM + SOY) and (ARS) diets performed better than the (CAN + EFA-based diet as the growth rates and average weights were significantly higher in fish fed with these two diets than those fed with (CAN + EFA) based diets at the end of the 8 weeks rearing trial. It was suggested that if a sufficient EFA is added to a (CAN + EFA-based diet such that it meets the requirements for cobia, it may enhance the survival rate, growth rate, and energy content in the fillet. Therefore, the essential amino acid requirements for cobia need to be determined to effectively utilize plant-based protein by balancing the nutrient with amino acid supplements like taurine, DL-methionine, L-lysine, L-threonine, L-tryptophan, L-isoleucine, and L-valine. Among the few determined optimal amino acid levels in the percentage of dry diet were methionine 1.19 %, cysteine 0.67 % [64], lysine 2.33 %, [65] and arginine 2.82 % 66].
Trushenski, et al. [67] reported that fish meal can be replaced with soya bean in cobia feed up to 50 % and above without reducing the growth, as well as feed intake but the final weight gain was only affected when fish oil was completely replaced. Therefore, among the identified factors limiting the inclusion of plant-based protein in cobia feeds at a higher level, including low protein content, low palatability, and the presence of anti-nutritional factors that reduce the digestion, absorption, and utilization of protein and amino acids. Craig [68] established that soy protein concentrate can effectively replace fish meal in cobia feed up to 75 % for juvenile cobia and in the presence of amino acid supplement, especially taurine, and that total replacement is possible without detrimental impacts on production characteristics. A similar scenario has been identified for other marine carnivorous species like Atlantic salmon (Salmo salar ) as they do not tolerate high levels of most plant protein feed ingredients in their diet [69]. It is through amino acid supplementation that efficient use of plant protein can be achieved in cobia production. For example, Silva, et al. [70] showed that juvenile Senegalese sole (Solea senegalensis) fed diets that contain 37 % fishmeal showed comparable growth with fish fed a 5 % fishmeal diet when essential amino acids were supplemented.
Poultry by-product Meal (PBM) is another good alternative protein source for cobia. Its potential to replace fishmeal totally without an adverse effect on growth has been confirmed, even though 60 % replacement was found to be optimal. Saadiah, et al. [71] fed juvenile cobia with fishmeal replaced with PBM at 20, 40, 60, 80, and 100 % dietary protein levels. At the end of the eight-week feeding trial, weight gains, specific growth rate, FCR, and protein efficiency ratio were not significantly different between the control and treatments. The best specific growth rate was recorded for fish fed with 60 % PBM. A similar, observation was recorded by Zhou, et al. [72] where the replacement of juvenile cobia fed with PBM up to 60 % showed no significant differences in biochemical and hematological parameters of the tested fishes. Therefore, enrichment of plant-based protein with amino acids to a balanced proportion required by cobia in various life stages will be essential for the utilization of some of these alternative proteins in cobia production.
Feed utilization in low salinity conditions
The culture of cobia in the pond from larvae to the juvenile stage has been successfully conducted using sea water [73]. Atwood, et al. [74] utilized a gradual reduction of (2 g.l-1.day) in salinity from 20 g.l-1 to test the resistance of juvenile cobia to low salinity. Mortality was recorded as the salinity approached 8 g.l-1 and nearly all fish died at 2 g.l-1 salinity level. Environmental influence on fish physiology is a complex phenomenon and salinity shift resulted in differential utilization of feed and disease response. Denson, et al. [48] reported that the growth rate of cobia was reduced significantly as salinity fell to 15 ppt and salinity reduction to 5 ppt resulted in poor health in cobia juveniles. Contrary to these findings, Resley, et al. [12] found that juvenile cobia held in salinity of 5 ppt., exhibited the same growth or better than the fish held in salinities of 15 and 30 ppt. as no significant differences were observed in the mean, weight gained specific growth rate and feed efficiency. Denson, et al. [48] fed the fish with a floating pelleted diet containing 44 % protein and 20 % lipid while Resley, et al. [12] fed the fish to satiation twice daily with feed containing 53 % crude protein and 13 % lipid. High protein feed may have enhanced the performance of cobia in low salinity. Holt, et al. [41] concluded that studies on cobia salinity tolerance suggest that the possibility of rearing cobia may be successful in salinities as low as 15 g.l-1 with the use of supplemented feeds. This is a possible explanation for the contrasting reports of Denson, et al. [48] and Resley, et al. [12]. Since the feeding regime doesn’t affect cobia growth performance [75], pond culture of cobia may become a reality in the future if other factors like feeding intensity and optimal diet composition in lower salinity are well understood. Pond culture is known to be advantageous in that fish may utilize natural food in the ponds.
Conclusion
As cobia farming develops worldwide through mariculture and other technologies, the diverse bottlenecks in cobia aquaculture have to be overcome. The use of genetic markers for trait selection like disease resistance, survival ability, and food utilization efficiency will be essential interventions for the improvement of cobia culture. An ability to utilize a wide range of plant and animal protein as food demonstrated by cobia signifies its promising future. Hence optimizing its inclusion with the required amino acid supplement is recommended for cost-effective production. Further understanding of larvae diet, especially for the fatty acid requirement, can be utilized to supplement life feed such that it reaches an optimal level required by cobia larvae. For the sustainability of the cobia aquaculture industry, improvements in the management of feeding strategies and a detailed understanding of the physiological basis of feed utilization in a low-salinity environment are crucial.
- Briggs JC. Fishes of worldwide (circumtropical) distribution. Copeia. 1960; 3: 171-180.
- Shaffer RV, Nakamura L. Synopsis of Biological Data on the Cobia Rachycentron canadum (Pisces: Rachycentridae) FAO Fisheries Synopsis. 1989; 153:3.
- FAO (Food and Agricultural Organization of the United Nations) (2021) Capture statistics of cobia in some of the important fishing nations from 2017 to 2021 https://wwwfaoorg/fishery/statistics-query/en/capture/capture_quantity
- Rohit P, Bhat US. Fishery and diet composition of the cobia R canadum (Linnaeus 1766) exploited along Karnataka coast Indian Journal of Fisheries. 2012; 59: 61-65.
- Meyer GH, Franks JS. Food of cobia Rachycentron canadum from the northcentral Gulf of Mexico Gulf Research Report. 1996; 9: 161-167.
- Franks JSN, Garber K, Warren JR. Stomach contents of juvenile cobia R. canadum from The Northern Gulf of Mexico Fisheries Bulletin. 1996; 94: 374-380.
- Smith JW. Life history of cobia Rachycentron canadum (Osteichthyes: Rachycentridae) in North Carolina waters Brimleyana. 1995; 23: 1-23.
- Liao IC, Huang T, Tsai W, Hsueh C, Chang S, Leano EM. Cobia culture in Taiwan: current status and problems Aquaculture. 2004; 237: 155-165.
- Chou RL, Su MS, Chen HY. Optimal dietary protein and lipid levels for juvenile cobia R. canadum. Aquaculture. 2001; 193: 81-89.
- Ganga U, Pillai NGK, Akhilesh KV, Rajoolshanis CP, Beni N, Hashim M, Prakasan D. Population dynamics of Cobia R canadum (Linnaeus 1766) off Cochin coast south-eastern Arabian Sea Indian Journal of Fisheries. 2012; 59: 15-20.
- Van der Velde TD, Griffiths SP, Fry GC. Reproductive biology of the commercially and recreationally important cobia Rachycentron canadum in northeastern Australia Fisheries Science. 2010; 76:33-43.
- Resley MJ, Webb KA Jr, Holt GJ. Growth and survival of juvenile cobia Rachycentron canadum at different salinities in a recirculating aquaculture system Aquaculture. 2006; 253: 398-407.
- Benetti DD, Orhun MR, Sardenberg B, Hanlon BO, Welch A, Hoenig R, Zink I, Rivera JA. Advances in hatchery and grow-out technology of cobia R. canadum (L). Aquaculture Resources. 2008; 39: 701-711.
- Gopakumar G, Abdul Nazar AK, Tamilmani G, Sakthivel M, Kalidas C, Ramamoorthy N, Palanichamy S, Ashok V, Maharshi V, Rao KS, Rao GS. Broodstock Development and Controlled Breeding of Cobia Rachycentron canadum Linnaeus 1766) from Indian Seas. Indian Journal of Fisheries. 2011; 58: 27-32.
- Hassler WW, Rainville RP. Techniques for hatching and rearing cobia Rachycentron canadum through larval and juvenile stages North Carolina Sea Grant Publication UNC-SG-75-30: 26. 1975.
- FAO (Food and Agricultural Organization of the United Nations) (2014) Cultured Aquatic Species Information Programme Rachycentron canadum (Linnaeus 1766) http://wwwfaoorg/fishery/culturedspecies/Rachycentron canadum/en.
- FAO (Food and Agricultural Organization of the United Nations) (2021) Global aquaculture production of cobia from 2017 to 2021 https://wwwfaoorg/fishery/statistics-query/en/aquaculture.
- Global Aquaculture Advocate. the global magazine for farmed seafood. 2014; 17(6): 6-7.
- Nhu VC, Nguyen HQ, Le T, Tran MT, Sorgeloos P, Dierckens K, Reinertsen H, Kjørsvik E, Svennevig N. Cobia Rachycentron canadum aquaculture in Vietnam: recent developments and prospects Aquaculture. 2011; 315: 20-25.
- Weirich CR, Smith TIJ, Denson MR, Stokes AD, Jenkins WE. Pond culture of larval and juvenile cobia Rachycentron canadum in the southeastern United States: initial observations Journal of Applied Aquaculture. 2004; 16: 27-44.
- Babatunde TA, Amin SMN, Romano N, Yusoff FM, Arshad A, Esa YB, Ebrahimi M. Gonad maturation and spawning of cobia, Rachycentron canadum (Linnaeus, 1766) off the Dungun coast, Malaysia. Journal of Applied Ichthyology. 2018; 1–8.
- Xan L. Advances in the seed production of Cobia Rachycentron canadum in Vietnam Marine Finfish Aquaculture Network. 2005; 1:5-8.
- Brown-Peterson NJ, Overstreet MR, Lotz JM, Franks JS, Burns KM. Reproductive biology of cobia, Rachycentron canadum, from coastal waters of the southern United States. Fisheries Bulletin. 2001; 99: 15-28.
- Lotz JM, Overstreet, RM, Franks JS. Gonadal maturation in the cobia, Rachycentron canadum , from the north-central Gulf of Mexico. Gulf Research Reports. 1996; 9:147-159.
- Fry GC, Griffiths SP. Population dynamics and stock status of cobia Rachycentron canadum caught in Australian recreational and commercial coastal fisheries Fisheries Management Ecology. 2010; 17: 231-239.
- Gold JR, Giresi MM, Mark A, Renshaw JCG. Population genetic comparisons among cobia from the northern Gulf of Mexico US Western Atlantic and Southeast Asia North American Journal of Aquaculture. 2013; 75: 57-63.
- Darden TL, Walker JM, Brenkert K, Justin R, Yost JR, Denson MR. Population genetics of Cobia ( Rachycentron canadum ): implications for fishery management along the coast of the southeastern United States. Fisheries Bulletin. 2014; 112: 24–35.
- Pruett CL, Saillant E, Renshaw M. Microsatellite DNA markers for population genetic studies and parentage assignment in cobia Rachycentron canadum Molecular Ecology. 2005; 5: 84-86.
- Phinchongsakuldit JP, Chaipakdee J, Collins JF, Jaroensutasinee M, Brookfield JFY. Population genetics of cobia ( Rachycentron canadum ) in the Gulf of Thailand and Andaman Sea: fisheries management implications Aquaculture International. 2013; 21: 197–217.
- Chu KB, Abdulah A, Abdullah SZ, Bakar RA. A Case Study on the Mortality of Cobia ( Rachycentron canadum ) Cultured in Traditional Cages. Trop Life Sci Res. 2013 Dec;24(2):77-84. PMID: 24575250; PMCID: PMC3935374.
- Ninh NH, Thoa PN, Knib W, Nguyen NH. Selection for enhanced growth performance of Nile tilapia (Oreochromis niloticus) in brackish water (15–20 ppt) in Vietnam Aquaculture. 2014; 428: 1–6.
- Atwood HL, Tomasso JR, Webb K Gatlin DM. Low-temperature tolerance of Nile tilapia, Oreochromis niloticus: effects of environmental and dietary factors. Aquaculture. 2003; 34: 41–251.
- Arnold CR, Kaiser JB, Holt JG. Spawning of cobia (R. canadum) in captivity. Journal of the World Aquaculture Society. 2002; 33: 205-208.
- Kaiser JB, Holt GJ. Species profile Cobia Southern Regional Aquaculture Center and the United States Department of Agriculture. 2005; (7202): 6.
- Stieglitz JD, Benetti DD, Hoenig RH, Sardenberg B, Welch AW, Miralao S. Environmentally conditioned year-round volitional spawning of cobia ( Rachycentron canadum ) in broodstock maturation systems Aquaculture Research. 2012; 43: 1557-1566.
- Franks JS, Ogle JT, Lob JM, Nicholson LC, Barnes DN, Larsen KM. Spontaneous spawning of cobia Rachycentron canadurn induced by human chorionic gonadotropin (HCG) with comments on fertilization hatching and larval development Proceeding of Gulf and Caribbean Fisheries Institutes. 2001; 52: 598-609.
- Faulk CK, Holt GJ. Biochemical Composition and quality of captive-spawned Cobia Rachycentron canadum eggs. Aquaculture. 2008; 279: 70–76.
- Weirich CR, Stokes AD, Smith TI, Jenkins WE, Denson MR. Outdoor tank and pond spawning of cobia, Rachycentron canadum in coastal South Carolina. Journal of Applied Aquaculture. 2006; 18(3):1-6.
- Nhu VC. Present Status of Hatchery Technology for Cobia Rachycentron canadum in Vietnam Aquaculture Asia Magazine Oct-Dec: 32–35. 2005.
- Sahoo SK, Giri SSS, Mohapatra CBC. Evaluation of breeding performance of Asian catfish Clarias batrachus at different dose of HCG and latency period combinations Turkish Journal of Fisheries and Aquatic Sciences. 2008; 8: 249-251.
- Holt GJ, Faulk CK, Schwarz MH. A review of the larviculture of cobia Rachycentron canadum a warm water marine fish Aquaculture. 2007; 268: 181-187.
- Philipose KK, Loka J, Sharma SRK, Divu D, Rao KS, Sadhu N, Dube P, Gopakumar G, Rao S. Farming of cobia Rachycentron canadum (Linnaeus 1766) in open sea floating cages in India Indian Journal of Fisheries. 2013; 60: 35-40.
- Benetti DD, Hanlon BO, Rivera JA, Welch AW, Maxe YC, Orhun MR. Growth rates of cobia ( Rachycentron canadum ) cultured in open ocean submerged cages in the Caribbean. Aquaculture. 2010; 302: 195-201.
- Faulk CK, Benninghoff AD, Holt GJ. Ontogeny of the gastrointestinal tract and selected digestive enzymes in cobia Rachycentron canadum . Journal of Fish Biology. 2007; 70: 1-17.
- Hitzfelder GM, Holt GJ, Fox JM, McKee DA. The effect of rearing density on growth and survival of cobia Rachycentron canadum larvae in a closed recirculating aquaculture system. Journal of the World Aquaculture Society. 2006; 37: 204-209.
- Huang CT, Miao S, Nan FH, Jung SM. Study on regional production and economy of cobia Rachycentron canadum commercial cage culture Aquaculture International. 2011; 19: 649–664.
- Weirich CR, Wills PS, Baptiste RM, Riche MA. Production characteristics and body composition of juvenile cobia fed three different commercial diets in recirculating aquaculture systems North American Journal of Aquaculture. 2010; 72: 43-49.
- Denson MR, Stuart KR, Smith TIJ, Weirich CR, Segars A. Effects of salinity on growth, survival, and selected hematological parameters on juvenile cobia Rachycentron canadum . Journal of the World Aquaculture Society. 2003; 34: 496–504.
- Sajeevan MK, Kurup BM. Evaluation of feeding indices of cobia Rachycentron canadum (Linnaeus 1766) from northwest coast of India Journal of the Marine Biological Association of India. 2014; 55 (2): 16-21.
- Somvanshi VS, Vargese S, Gulati DK, Bhargava AK. Some Biological aspects of kingfish Rachycentron canadum (Linnaeus 1766) from the north-west Indian EEZ Occasional Paper Fishery Survey of India Mumbai. 2000; 36.
- Arendt MD, Olney JE, Lucy JA. Stomach content analysis of cobia, R. canadum from lower Chesapeake Bay. Fisheries Bulletin. 2001; 99: 665-670.
- Nguyen HQ, Tran TM, Reinertsen H, Kjørsvik E. Effects of dietary essential fatty acid levels on broodstock spawning performance and egg fatty acid composition of cobia Rachycentron canadum Journal of the World Aquaculture Society. 2010; 41: 687-699.
- Buke E. Sea Bass (Dicentrarchus labtax L., 1781) seed production. Turkish Journal of Fisheries and Aquatic Science. 2002; 2: 61-67.
- Faulk CK, Holt GJ. Lipid nutrition and feeding of cobia Rachycentron canadum larvae Journal of the World Aquaculture Society. 2003; 34: 368-378.
- Rasdi NW, Jian GQ. Improvement of copepod nutritional quality as live food for aquaculture: a review Aquaculture Research. 2016; 47(1)1–20.
- Maehre HK, Hamre K, Elvevoll EO. Nutrient evaluation of rotifers and zooplankton: feed for marine fish larvae Aquaculture Nutrition. 2012; 19: 301-311.
- Drillet G, Frouel S, Sichlau MH, Jepsen PM, Højgaard JK, Joarder AK, Hansen BM. Status and recommendations on marine copepod cultivation for use as live feed. Aquaculture. 2011; 315:155–166.
- Benetti DD, Sardenberg B, Welch A, Hoenig R, Orhun MR, Zink I. Intensive larval husbandry and fingerling production of cobia R. canadum. Aquaculture. 2008; 281: 22-27.
- Ditty JG, Shaw RF. Larval development, distribution, and ecology of cobia R. canadum (Family: Rachycentridae) in the northern Gulf of Mexico. Fisheries Bulletin. 1992; 90: 668-677.
- Zink IC, Cavalin FG, Bacoat D, Denlinger B, Palmer K, Sardenberg B, Kirkpatrick R, Orhun R, Benetti D. US trials compare commercial diets fed to juvenile cobia. Global Aquaculture Advocate. 2006; 9(2):53-4.
- Lunger AN, Craig SR, McLean E. Replacement of fish meal in cobia ( Rachycentron canadum ) diets using an organically certified protein. Aquaculture. 2006 Jun 30;257(1):393-399. doi: 10.1016/j.aquaculture.2005.11.010. Epub 2006 Jan 10. PMID: 32287453; PMCID: PMC7112107.
- Nguyen MV, Rønnestad I, Buttle L, Lai HW, Espe M. Evaluation of a high plant protein test diet for juvenile cobia Rachycentron canadum in comparison to commercial diets. Journal of Agricultural and Crop Research. 2014; 2: 117-125.
- Watson AM, Barrows FT, Place AR. Taurine supplementation of plant derived protein and n-3 fatty acids are critical for optimal growth and development of cobia, Rachycentron canadum . Lipids. 2013 Sep;48(9):899-913. doi: 10.1007/s11745-013-3814-2. Epub 2013 Jul 25. PMID: 23884630.
- Zhou QC, Wu ZH, Tan BP, Chi SY, Yang QH. Optimal dietary methionine requirement for juvenile Cobia (Rachycentron canadum) Aquaculture. 2006; 258: 551-557.
- Zhou QC, Wu ZH, Chi SY, Yang QH. Dietary lysine requirement of juvenile cobia ( Rachycentron canadum ) Aquaculture. 2007; 273: 634-640.
- Ren M, Ai Q, Mia K. Dietary arginine requirement of juvenile cobia ( Rachycentron canadum ) Aquaculture Research. 2014; 45: 225-233.
- Trushenski JT, Laporte J, Lewis H, Schwarz M, Delbos B, Takeuchi R, Sampaio LA. Fish meal replacement with soy-derived protein in feeds for juvenile cobia: influence of replacement level and attractant supplementation Journal of the World Aquaculture Society. 2011; 42: 435-443.
- Craig SR. Soy use optimization in feeds for cobia. American Soybean Association-International Marketing Technical Bulletin AQ. 2012; 50: 2.
- Gatlin DM, Barrows FT, Brown P, Dabrowski K, Gaylord TG, Hardy RW, Herman E, Hu G, Krogdahl A, Nelson R, Overturf K. Expanding the utilization of sustainable plant products in aquafeeds: a review Aquaculture Research. 2007; 38(6): 551-579.
- Silva JMG, Espe M, Conceiçao LEC, Dias J, Valente LMP. Senegalese sole juveniles ( Solea senegalensis Kaup 1858) grow equally well on diets devoid of fish meal provided the dietary amino acids are balanced Aquaculture. 2009; 296(3): 309-317.
- Saadiah I, Abol-Munafi AM, Che Utama CM. Replacement of fishmeal in cobia ( Rachycentron canadum ) diets using poultry by-product meal Aquaculture International. 2011; 19: 637-648.
- Zhou QC, Zhao J, Li P, Wang HL, Wang LG. Evaluation of poultry by-product meal in commercial diets for juvenile cobia ( Rachycentron canadum ) Aquaculture. 2011; 322-323: 122–127.
- Benetti DD, Orhun MR, Zink I, Cavalin FG, Sardenberg B, Palmer K, Denlinger B, Bacoat D, O'Hanlon B. Aquaculture of cobia ( Rachycentron canadum ) in the Americas and the Caribbean. In: Liao, I.C., Leano, E.M. (Eds.), Cobia Aquaculture: Research, Development, and Commercial Production. Asian Fisheries Society, Keelung, Taiwan 2007; 10.
- Atwood HL, Young SP, Tomasso JR, Smith TTJ. Resistance of cobia, Rachycentron canadum , juveniles to low salinity, low temperature, and high environmental nitrite concentrations. Journal of Applied Aquaculture. 2004; 15: 191-195.
- Costa-Bomfim CN, Pessoa WVN, Oliveira RLM, Farias JL, Domingues EC, Hamilton S, Cavalli RO. The effect of feeding frequency on growth performance of juvenile cobia, Rachycentron canadum (Linnaeus, 1766). Journal of Applied Ichthyology. 2014; 30: 135–139.
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.
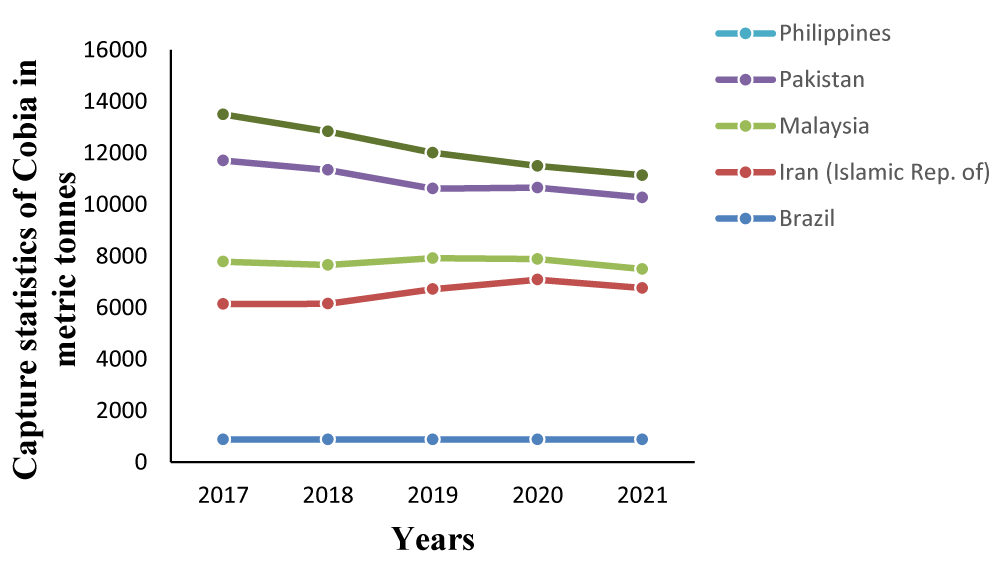


 Save to Mendeley
Save to Mendeley
