Assessment of ecological risk for ecosystems caused by dredged materials in freshwater environments
1Department of Biology, Polydisciplinary Faculty of Khouribga, University Sultan Moulay Slimane, Khouribga, Morocco
2Laboratory of Life Science and Earth (SVT), Regional Centers for the Professions of Education and Training (CRMEF), El Jadida, Morocco
Author and article information
Cite this as
Salama Y, Chennaoui M (2024) Assessment of ecological risk for ecosystems caused by dredged materials in freshwater environments. Glob J Ecol. 2024; 9(1): 030-041. Available from: 10.17352/gje.000094
Copyright License
© 2024 Salama Y, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Several chemical substances are released into the environment, and many of them accumulate in the silt or sludge constituting sediments, at the bottom of rivers and lakes as well as in estuaries and seabeds. The ecological and health damage caused by the contamination of sediments by these chemical substances can have serious consequences on ecosystems by reducing the diversity of communities, particularly invertebrates, with indirect consequences on fish populations, loss of edibility of fish or shellfish, recreational uses affected or impossible, ecological risk assessment can be used prospectively or retrospectively. The general outline of the evaluation includes three stages: the formulation of the problem, which aims to develop an analysis plan and define the means to be implemented to characterize the risk, the analysis itself, where we collect the data necessary for the assessment of effects and exposure, and risk characterization, where the risk(s) and, as far as possible, the associated uncertainties are estimated. The application of this approach to dredged materials is proposed in the form of two successive stages: the first consists of a simplified risk analysis, from which we can decide to initiate a second stage of detailed evaluation. The review evaluates the ecological risk associated with the disposal of dredged materials in freshwater environments. It examines the impact of dredged materials on biodiversity and ecosystem health, focusing on assessing the extent of ecological damage, identifying influential factors, and proposing mitigation strategies.
Among the chemical substances released into the environment, many of them accumulate in silt or mud making up sediments, at the bottom of rivers and lakes as well as in estuaries and oceans [1,2]. Many of the contaminated sediments were polluted years ago, by pollutants that are now often almost absent from surface waters. The sometimes very long-term persistence of these pollutants in sediments can cause adverse effects on both the ecosystem and human health. The ecological and health damage caused by sediment contamination has a real social cost [3]: this is the reduction in the diversity of communities, particularly of invertebrates, with indirect consequences on fish populations, loss of edibility of fish or shellfish, recreational uses affected or impossible, and additional costs linked to the management of contaminated materials during dredging or cleaning operations. The most frequently cited contaminants are metals (cadmium, chromium, copper, mercury, nickel, lead, and zinc), arsenic, Polychlorinated Biphenyls (PCBs), and polycyclic aromatic hydrocarbons (PAHs) [4,5]. Deposits of sediment, contaminated or not, can lead to cleaning needs when they increase the risk of flooding, or reduce the draft of navigable parts [6,7]. The problems of contamination of extracted materials, encountered by all industrialized countries, very early on led to the development of evaluation procedures and methods, subsequently enabling management decisions to be made [8-10].
The management of these problems of contamination of sediments and dredged materials is part of the general context of risk management; this always goes through – formally or not – through an evaluation stage, which may not be decisive in itself in the decision-making process [11,12], depending in particular on the degree of uncertainty inherent in any evaluation. of risk. The US EPA (U.S. Environmental Protection Agency) has made ecosystem risk assessment a major focus of its multi-year ecological research program [13]. Through field studies (“place-based research”), it seeks in particular to develop methods for assessing risks in situations of multiple contaminations, or even the conjunction of “stressors” of different natures (physical, biological, and chemical) [14].
The risks targeted by both assessment and management concern on the one hand man, his health and his activities, and on the other hand ecosystems. These two aspects are generally considered separately, notably for practical reasons [15]. The assessment of exposure depends on knowledge of transfers from the “source term” (the contaminated sediment) to the target(s), humans in one case, and one or more ecosystems in the other. In any case, the phenomena at stake are the same in both cases: it will involve the remobilization of contaminants, their biotic or abiotic transformations, water transport or accumulation via food chains [16]. The main difference between these two types of evaluation approaches therefore lies at the level of the “targets”, which are more numerous and diversified in the case of ecosystems. This intrinsic complexity may seem prohibitive at first glance, and lead to avoiding tackling the assessment of ecological risk.
In absolute terms, it would naturally be desirable for the management decisions ultimately taken to integrate the “health” and “ecosystems” aspects of the evaluation, but neither the conceptual framework nor the “tools” currently seem ready for this integration. A specific guide for the assessment of health risks generated by contaminated sediments is being prepared under the aegis of the “The Competence Center – Polluted Sites & Soils” of Nord-Pas-de-Calais [17]. Ecological risk assessment can be used prospectively or retrospectively, and in some cases, both aspects can be addressed in the same study (for example, this type of approach can be used to identify the cause of the decline of a population and evaluate the effects of corrective measures) [18]. It can also be used to compare different corrective solutions or to establish priorities in the case of complex situations [19].
The purpose of this review is to assess the ecological risks posed to freshwater ecosystems by the disposal of dredged materials. The study aims to quantify the potential impact on biodiversity, water quality, and overall ecosystem health resulting from the presence of dredged materials. By evaluating these risks, the study intends to provide valuable insights for decision-making processes regarding dredging activities in freshwater environments.
Methodological approach for ecological risk assessment
General methodological framework
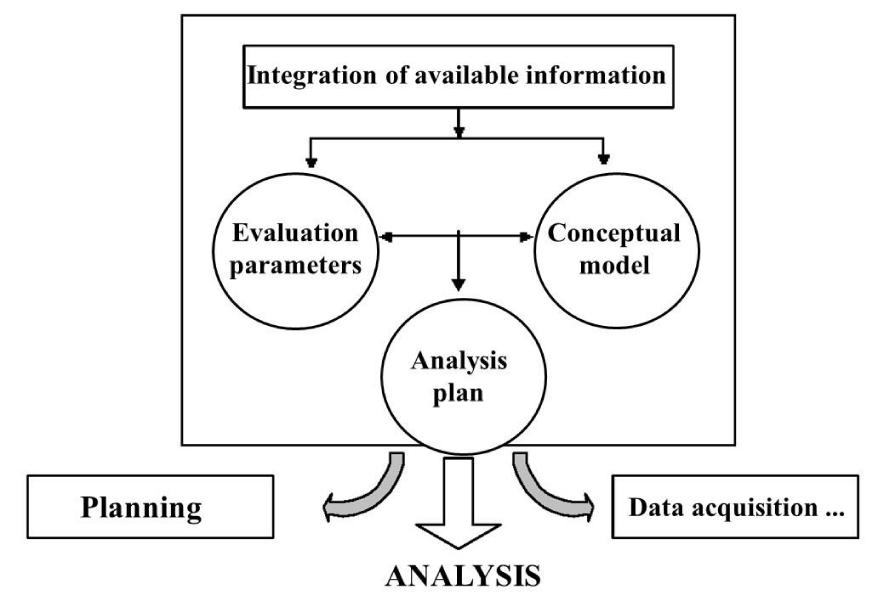
The general diagram, taken up by most authors [8,20,21], includes three stages (Figure 1): problem formulation, which aims to develop an analysis plan and define the means to be implemented to characterize the risk, the analysis itself, where we collect the data necessary for the evaluation of effects and exposure, and the characterization of risks, where we estimate the risks and, as far as possible, the associated uncertainties. This process is part of the broader and more complex process of risk management, of which only certain stages are mentioned in Figure 1. The linear representation adopted for convenience is not necessarily consistent with reality, which can admit iterations, especially during the problem formulation phase. Likewise, certain management plans provide for evaluations at several levels (simplified then detailed evaluations).
Data acquisition takes place at different levels of the process; it may be pre-existing data or produced for the occasion at the problem formulation or analysis stage. The characterization of risk, and even more so its subsequent management, gives rise to the acquisition of data aimed at verifying forecasts and the effect of the measures taken.
Presentation of the different stages
Problem formulation: This first stage of the evaluation process is a critical stage, the objective of which is to frame the analysis and characterization phases, by precisely identifying the data to be acquired, the measurement or evaluation techniques, and the framework. interpretation. This phase essentially includes three steps [22]: (1) a detailed description of the context and integration of available data, (2) selection of “evaluation parameters” and development of the conceptual model, and (3) development of an analysis plan (Figure 2).
The “evaluation parameters” are a formal expression of what we want to protect or evaluate in the ecosystems concerned [23,24]. They make it possible to make the link between the management objectives to be the origin of the triggering of the evaluation and the measurements and analyses (or “measurement parameters”) to be implemented to carry it out. They, therefore, represent elements of the ecosystem likely to be affected by the factor(s) of risks studied, without necessarily being directly measurable [25,26]. Their formulation must be precise, and they must also facilitate communication with the parties interested in the evaluation, by putting as much emphasis on as possible focus on the elements of the ecosystem that are of value to these parties.
The conceptual model is a series of hypotheses based on the relationships between the sources of the “stress” studied, the effects of this stress, and the effect parameters (endpoints) [27]. It can be based on scenarios, in particular when it comes to generic evaluations, that is to say not linked to a specific space. This is particularly the case for chemical substances or for the assessment of the eco-compatibility of waste storage or recovery scenarios [24,28-30]. This notion of scenario can be extended to other areas, as long as there is some interest in standardizing approaches.
Analysis phase: It consists of the acquisition of the data necessary to characterize the effects of pollutants and the exposure of the different targets concerned. The different evaluation tools resulting from the formulation of the problem that can be implemented can be classified into two main categories [24]:
− Experimental models,
− Mathematical models, including statistical models and deterministic models.
The experimental models aim to reproduce field conditions as best as possible and to observe the consequences of the introduction of pollution on ecosystems. Their main advantage lies in the realism of the observation carried out (only for the duration tested, however) and their main disadvantages lie in the cost and time of the studies carried out, in the difficulties encountered in controlling all the parameters of complex experimental devices. implemented, as well as in the "black box" side of the work carried out when it is not accompanied by a parametric study of the observed phenomena. The consequence of this "black box" aspect is the impossibility of making long-term projections based on the observations made. Statistical models require a lot of data and are therefore not easily used in operational approaches like the one described in this report; These are more research tools, usable on heavily instrumented sites [31,32].
The deterministic models are developed after a detailed analysis of the physical, chemical, and biological phenomena that can occur during the exposure, under the conditions of the scenario, of the target ecosystems to the polluted materials studied. The prediction of these phenomena can be based on theoretical data, but it can also, and this is in our opinion entirely desirable given the complexity of the phenomena involved in the case of sediment deposits, be based on experimental results. Concretely, the analysis of effects will be based on biological approaches, which mainly include bioassays and bioindicators [33-35].
Exposure assessment consists, in principle, of determining the probabilities of contact between the causal factor (stressor) and the “targets” (receptors) [24]. It therefore involves the analysis of sources, transfers from these sources, and the distribution of the contaminant in the environment. This analysis can be carried out using theoretical calculations (water balance of the site based on data from the hydrogeological study of the site, for example) as well as on the basis of experimental results (column tests to evaluate the transfer pollutants in the context of the water balance, for example).
Risk characterization phase: The characterization of risks results from comparing the assessment of effects with that of exposure; very generally speaking, there are a variety of possible methods, of varying complexity. The choice will depend:
− Operational constraints (summary or detailed assessment, deadlines, resources, etc.)
− Available data (nature and number).
The following are thus proposed as methods of characterizing or estimating risks:
− Field studies (subject, among other things, to establishing a causal link): here we can cite the case of the poisoning of passerines by carbofuran [23,36]. This type of approach will not apply to evaluations with predictive purposes.
− Categorization, and classification: these are approaches of a qualitative nature, but which nevertheless allow the use of mathematical tools. They can be used for comparative approaches (for example, comparison of two decontamination methods), for which it is not necessary to have an absolute answer.
− Quotient method: in this case, we reduce the estimate of exposure and that of danger to point values, possibly associated with an uncertainty range.
− Comparisons integrating all stressor/response relationships: in this case, the aim is to estimate the level of risk associated with a given level of exposure.
Quotient method: In this approach, each of the two components of risk (danger, exposure) is represented by a single concentration; the value of the exposure/danger quotient makes it possible to summarily determine the level of risk (if this quotient is greater than 1, the risk is considered significant). The exposure component is commonly referred to by the acronym “PEC” (Predicted Environmental Concentration); it can be estimated by calculation, based on knowledge of the source term and the methods of transfer into the environment. It can also be determined from measurement data, in this case, the PEC will correspond to a statistical indicator (average, median in particular) [22]. When several compartments (water, sediment, etc.) are examined, it is important to determine a PEC for each compartment. The danger component is designated by the acronym “PNEC” (Probable No Effect Concentration). This is determined from the results of bioassays, in principle covering several trophic levels. Safety or extrapolation factors are applied to the lowest values, so as to take into account variations in sensitivity between species (i.e. it is not necessarily the most sensitive species that were tested), or within the same species. In other cases, for example in the absence of chronic toxicity data, an extrapolation factor will be applied to the acute toxicity data [35]. The factor ultimately applied is therefore a function of the available data set. Extrapolation factors may vary depending on regulatory or other contexts; Table 1 presents commonly used factors and the corresponding minimum data sets. When several compartments (water, sediment, etc.) are examined, it is important to determine a PNEC for each compartment [35].
These safety factors, or extrapolation factors, ultimately aim to formalize conservative choices in situations where uncertainty is typically significant [37,38], since it involves, for example, defining a level of risk-free concentration for freshwater aquatic species based on data on 3 species under laboratory conditions. Garcia-Reyero & Murphy, 2018 [37], while adopting a generally reserved, even critical, attitude on the use of these factors, nevertheless show, on several examples, that they can effectively make it possible to embrace the range of interspecific sensitivities (they cite for example the case of data on mammals from the Great Lakes, for which 50% of the LD50 are included in a range from x1 to x4, and 96% at x100).
This quotient approach is currently very widely used, particularly in the regulatory framework for the risk assessment of chemical substances, or simplified risk assessments [39]. The determination of PNEC is also the method most frequently implemented in the process of developing quality criteria for water [40-42].
Comparing distributions: When we really want to have a quantitative assessment, and when the data allows it, it is possible to proceed by comparing distributions, concentrations of toxic effects on the one hand, and exposure concentrations on the other. This type of approach makes it possible to estimate a risk probability for an exposure concentration level, associated with a confidence interval [43,44]. This possibility will be particularly useful in the case where we want to test several risk reduction possibilities or estimate the residual risks according to the level of intervention. The different practical possibilities amount to comparing a dose-response curve, or a distribution of effect values, to a cumulative distribution of exposure concentrations [2,45].
Risk assessment for dredged products
The environmental impacts of dredged products, like other materials, have been known for many years, and have gradually led to the establishment of evaluation procedures, which do not always refer explicitly to the risk assessment [46]. However, it seems that the introduction of risk assessment approaches is a logical evolution of the conformity assessment procedures of dredged products [47]. In the marine environment, the management of dredged products mainly falls under the London Convention (1972) on the prevention of marine pollution by depositing waste and other materials [48,49]. Deposits of dredged products, as well as sewage sludge, fish residues, etc., and wrecks, can be deposited at sea subject to authorization, following an assessment of their safety.
Applications
The step-by-step strategy has been applied for a long time to the problems of managing dredged products, without always making explicit reference to a risk assessment framework: in this type of approach, proposed for the marine environment, waters fresh inland waters [50] and adapted to the North American Great Lakes [51], the aim is to optimize the assessment of the potential impacts of the deposit of dredged products in water or wetland. To do this, at each stage, the possibility of making a decision is examined on the basis of the available information, and the next stage, involving more sophisticated means of investigation, is only undertaken if the uncertainty is too big. More precisely, in the example of the Great Lakes, the 4 steps of the approach involve the following types of data:
- Criteria for exemption from the test procedure, the existence of historical data.
- Physico-chemical and benthic invertebrate data collection for this step, calculation by model of the worst conditions for the water column and bioaccumulation.
- Toxicity bioassays (6 recommended organisms: for the water column, the cladoceran crustaceans Daphnia magna, Ceriodaphnia dubia, and the fish Pimephales promelas; for the sediment, the insect Chironomus tentans, the amphipod crustacean Hyalella azteca and the oligochaete Lumbriculus variegatus).
- Specific approach, in practice no precise recommendation.
Although this type of approach does not explicitly claim to be an ecosystem risk assessment, it is nevertheless clear that it has at least some similarities, in particular the combination of biological methods and chemical analyses, as well as transfer tests. The main difference is that the conceptual model is implicit, as are the evaluation parameters. The risk characterization, not mentioned as such, is however carried out in a simple manner, the interpretation being centered on the quality criteria for the water column. Another criticism could be made, less of the approach itself, which appears rational and uses standardized tools, than of the management plan for dredged products, reduced to a single destination. This scheme seems too simple, or even generally inapplicable in the French case, where the available spaces are much less numerous and extensive.
In the French context, the management scenarios mentioned [52] currently include spreading, deposit on land with or without containment, landfill, recycling, and deposit underwater (clapage). There is currently a lack of a precise description of these management methods and their variants, a description essential to the problem formulation phase of risk assessments.
Another difficulty encountered here in this attempt to develop a procedure for assessing the ecological risk of dredged products is the absence of a standardized approach for the initial characterization of the sediments, which will also be used during the problem characterization phase. It would be desirable for this approach not only to be based on physical and chemical parameters but to include biological parameters, to gain relevance in the initial diagnosis.
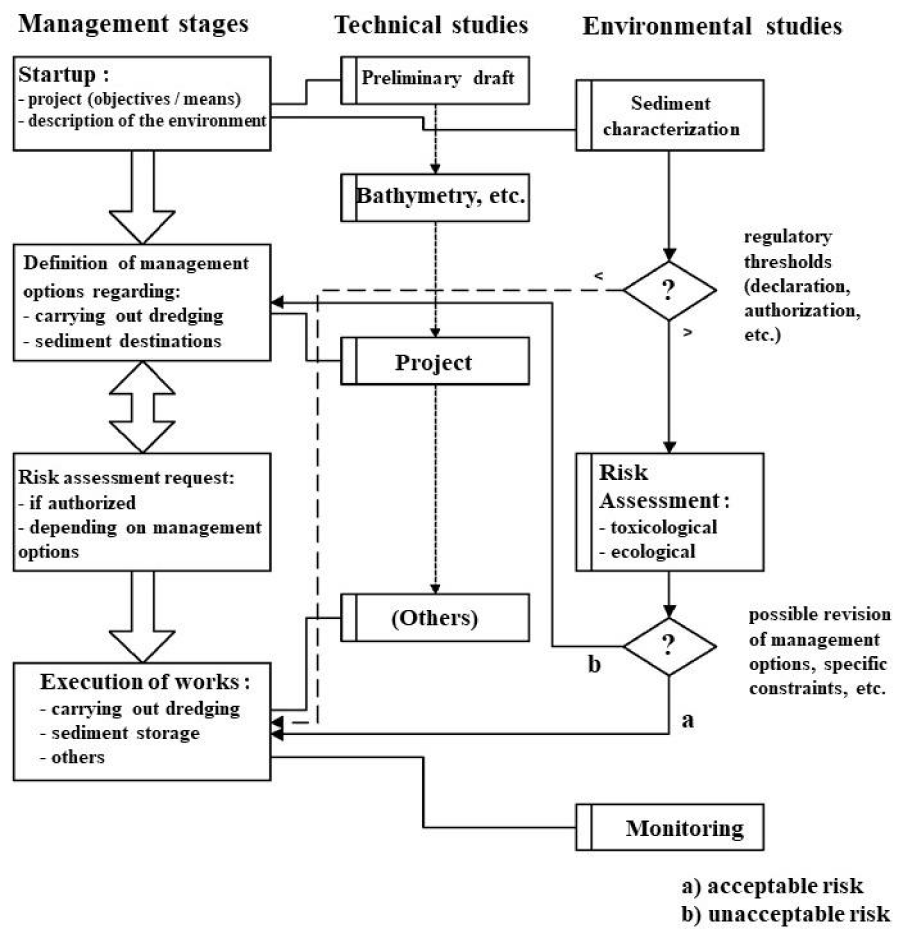
Figure 3 summarizes all the stages of a dredging project, including the initial characterization and the risk assessment in its two aspects. When the result of the evaluation leads to a risk that is too high in relation to the chosen criteria (see arrow b), it is appropriate to define one or more other management options and start the evaluation again, unless the data collected does not make it possible to characterize the risks for these new management options. Likewise, for a given management scenario, we can first carry out a “simplified assessment”, and then, if the risk is considered too high, move on to a detailed assessment as is particularly planned for contaminated sites [46,53].
Problem formulation
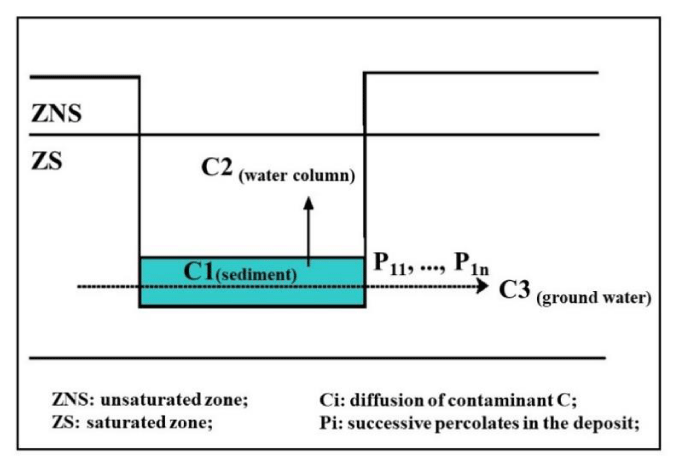
Gravel pit deposit: It is particularly recommended to backfill in successive bins (with daily updating of a plan of the deposit), proceeding from the hydraulic downstream towards the upstream. The piezometric level must not be disturbed by this backfilling, which excludes clogging materials. In all cases (regulations on classified installations or water law), the materials used for backfilling must be inert, that is to say, materials that “do not undergo any significant physical, chemical or biological modification, do not decompose, do not burn or produce any other chemical or physical reaction, are not biodegradable and do not damage other materials with which they come into contact (…). The total production of leachate and the pollutant content of the waste, as well as the ecotoxicity of the leachate, must be negligible and not undermine the good ecological quality of the water (…)” [54]. One of the techniques used consists of depositing the sediments in a water gravel pit. Figure 4 below corresponds to a schematic representation of this type of deposit.
Ecosystem concerned: Even if we do not have detailed faunal lists concerning the organisms present in gravel pits used for this type of deposit, the presence of a three-level food web is very probable, especially if the gravel pits are likely to accommodate the dredged sediments are sites frequented by fishermen. Therefore, we must consider as potential targets in the ecological risk assessment an ecosystem comprising [39]:
- Primary producers, phytoplankton, including uni- and multicellular green algae, possibly macrophytes,
- Primary consumers, pelagic and benthic micro and macroinvertebrates, in particular microcrustaceans and aquatic insects,
- And finally secondary consumers, including amphibians, fish, and aquatic birds feeding on invertebrates.
The presence of a 4th trophic level (fish for example, or piscivorous birds) is obviously probable. This level is subject to exposure to sediment contaminants through food, involving bioaccumulation phenomena [16]. For the moment, however, we do not plan to take this level(s) into consideration in the evaluation. It is not that we consider that the populations concerned are of less importance from an ecological point of view, but rather that the assessment of exposure, in the case of secondary poisonings, is much more complex, and that at the current stage of development of risk assessment procedures and their application in management issues, this complexity is not manageable. That said, this is clearly a subject that should not be neglected in future developments, especially as it has implications for health risks.
Transfer and transformation of contaminants: The main transfer routes pass through the aqueous phase (pore water then water column located above the sediment or water table). Initially, following the discharge of sediments into the gravel pit, the pollutants present in the pore water will be partially released into the water of the gravel pit and diluted therein. The contaminants present in the sediments can then, following dissolution/desorption phenomena, pass into the dissolved phase or be transferred in colloidal form. The main transfer mechanisms are then diffusion towards the surface water column and, taking into account the recommendations on the creation of the deposit, horizontal percolation in a saturated environment towards the lateral groundwater.
Secondarily, transfers of fine particles can also occur laterally from the surface of the deposit (bottom currents). Added to these mechanisms are the phenomena of physico-chemical or biological transformation. The pollutants concerned are those identified at the sediment characterization stage (Figure 3) as well as certain toxic natural constituents of the sediments, such as manganese, to the extent that it could be present in the water under anoxic and reducing conditions or after re-oxygenation [55,56], or ammonia [57,58].
Within the sediment, metallic contaminants can undergo modifications in terms of speciation (see among others [59,60]. Organic contaminants can be biodegraded. For contaminants like PAHs, biodegradation is in any case very slow and weak, particularly in anaerobic conditions, likely to occur in the lower layers of contaminated sediments [61,62], and with a disappearance especially of the compounds at lower molecular weight (approximately 15% in 50 days) [62,63]. Some recent work has focused on the photodegradation of certain PAHs, which makes them significantly more toxic, but this phenomenon does not seem very important for bottom sediments [64,65]. These general elements will be completed on a case-by-case basis, depending on the contaminants actually present in the sediment (Table 2).
The main route of transfer of contaminants is therefore linked to the circulation of water in the deposit: release of part of the pore water, then diffusion of pollutants towards the water column, as well as percolation of groundwater through the layer of deposited sediment. From the perspective of exposure assessment, transfer processes can be represented by ratios (dilution coefficients, mass/volume ratio), which is essential to determine the design of the analysis plan.
Exposure and effects models: The toxic effects on aquatic organisms or sediment organisms depend on the contaminants concerned; It will therefore be appropriate, in each case, to search for existing bibliographic data on the contaminants detected during the characterization phase. In the case of very ubiquitous contaminants, such as metals or PAHs. Furthermore, certain natural constituents of sediments, such as sulphides, can cause toxic effects on aquatic organisms [66,67]. This type of effect will occur particularly in the event of a sudden change in the structure of the sediment. Also, certain pollutants are likely to cause genotoxic effects [68,69]. If we do not yet know what additional risk this type of effect can generate, its potential consequences are such that they cannot be neglected, and that their consideration in the risk assessment procedures for the environment must be considered.
Exposure of organisms can be considered at the level of the water column, or during contact with the deposit. The management scenario studied therefore leads to an interest in these two environments:
− The sediment deposited in its entirety could: (1) affect the organisms pre-existing in the gravel pit, invertebrates, fish, or even birds [70], (2) limit possible recolonization due to its poor quality.
− The water is in contact with the sediment (sediment-water interface) in which the contaminants are likely to diffuse, as well as the pore water diffusing in the water column.
As a first step, we will favor standardized or normalized means of measurement, even if this means not testing all potential targets. This is the reason why the approach adopted for the evaluation of effects is based on bioassays on invertebrates, algae, and bacteria. The assessment of exposure also requires the precise definition of the mass/volume ratios involved:
− “Surface water volume/sediment volume” (VWS/VS) ratio;
− “Surface water volume/volume of pore water sediments released” (VWS/VWP) ratio; this ratio is used to determine the exposure of species in the water column above the deposit, and therefore to set the range of dilutions tested;
− “Groundwater volume/gross sediment mass passed through by percolation” (VWG/VSP) annual ratio; this ratio is used to calculate the concentrations in the water table, and therefore allows the volumes of percolates to be calibrated.
These ratios, essential to know for the analysis phase (conducting tests), are defined on the basis of a simplified water balance of the site, to be carried out during the problem formulation phase, in particular by means of a study hydrogeological.
Soil deposit
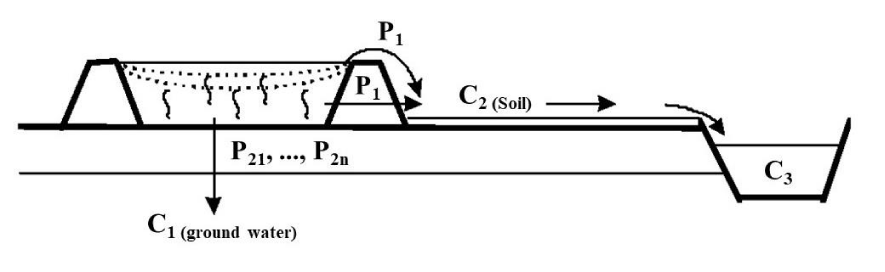
Regulatory and technical framework: When the landfill corresponds to a storage center for inert waste (class III landfills), the regulatory requirements that apply are those relating to this type of center. When the deposit on land corresponds to specific storage of sediments carried out within or outside the canal right-of-way, the regulatory requirements are less well defined. One of the main techniques used consists of making an unconfined deposit on the soil near or along the canal. Figure 5 below corresponds to a schematic representation of this type of deposit. If the repository is located far enough from the water's edge, certain targets no longer need to be taken into consideration, and the evaluation parameters must be adjusted accordingly.
− Ecosystems concerned: For this type of deposit, the main targets concerned are:The water table located under the deposit (target C1) which receives part of the percolates (P2) from the sediments. The concentration of these is potentially variable over time (P21, P21,…, P2n),
− The ground (C2) located near the deposit, the seat of flows coming from occasional overflows of the deposit, and the passage of sediment drainage water through the non-watertight dikes of the deposit (P1 percolates),
− And the aquatic environment (C3): canal or other watercourse located near the depot which receives the runoff water after runoff on the ground.
• Target C1 (Groundwater): The priority for this target concerns the protection of water resources. For C1, it is therefore proposed to only carry out a comparison of the expected concentrations in groundwater with the regulatory threshold values defined for potable water.
• Target C2 (Soil): Potential target organisms to consider are: plants: pioneer plants (chenopods, Polygonaceae, etc.) or meadow plants (ryegrass, etc.),/ the soil microflora (microflora of the soil itself and the rhizosphere), /and soil fauna including insects and earthworms.
• Target C3 (Canal or rivers): The trophic levels and potential targets to consider are primary producers, phytoplankton, including uni and multi-cellular green algae, / primary consumers, pelagic and benthic micro and macroinvertebrates, in particular microcrustaceans and aquatic insects, / and secondary consumers, including amphibians, fish and aquatic birds.
Transfer of pollutants: In this scenario, the main routes of pollutant transfer are through the aqueous phase. Taking into account the method of construction of the deposit, this transfer will be either lateral (by overflow of dripping water from the deposit or by passing through leaky dikes), or oriented from the deposit towards the water table. As for the “gravel pit” scenario, the transfer considered initially concerns the pollutants identified during the preliminary campaigns (trace elements, notably copper and zinc; polycyclic aromatic hydrocarbons (PAHs); polychlorinated biphenyls (PCBs)), as well as manganese and ammonia. Within the deposit, metallic contaminants can undergo modifications in terms of speciation; organic contaminants can be biodegraded. The biodegradation of PAHs, the main organic contaminants in the present case, will a priori be low given the probable anaerobic nature within the deposit and the short residence time during runoff on the ground. In any case, this potential degradation will mainly concern low molecular weight compounds. The main transfer processes associated with the “Deposit on Soil” scenario are listed in Table 3.
Exposure and effect model: The toxic effects on target organisms depend on the contaminants concerned; It will therefore be appropriate, in each case, to search for existing bibliographic data on the contaminants detected during the characterization phase. Exposure of organisms can be considered at the level of the soil and watercourses surrounding the repository. The management scenario studied therefore leads to an interest in these two environments subject to drainage water from the deposit which could: (1) affect the microflora, vegetation, and fauna of the soil; (2) affect the microflora, phytoplankton, and fauna of the watercourse. As a first step, we will favor standardized or normalized means of measurement, even if this means not testing all potential targets. This is the reason why the approach adopted for the evaluation of effects is based on bioassays on bacteria, higher plants, unicellular algae, aquatic invertebrates, and amphibians (genotoxicity).
The assessment of exposure also requires a very precise definition of the mass/volume ratios involved:
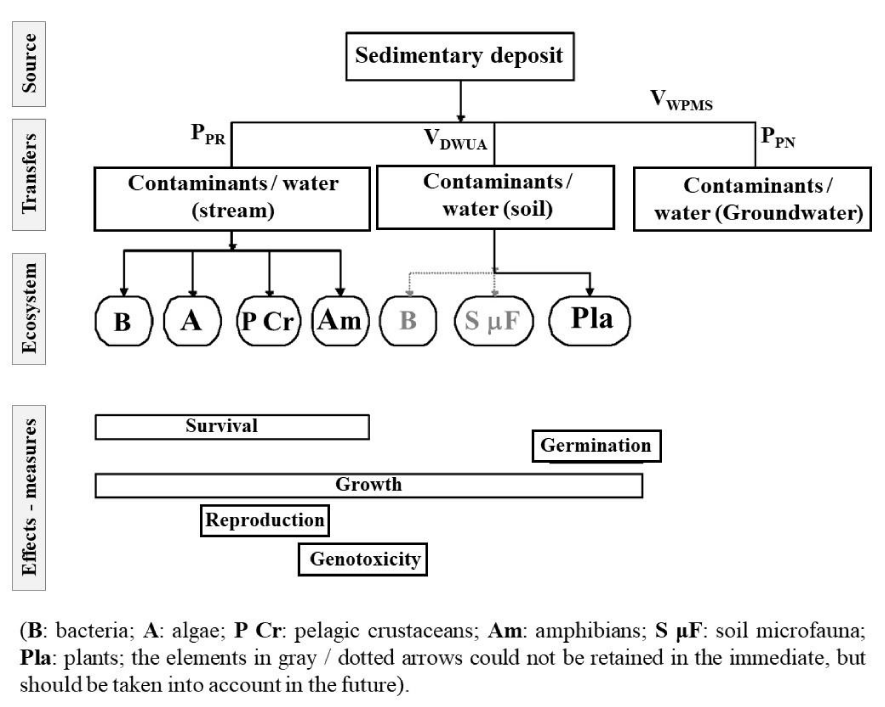
- Ratio “annual volume of dripping water per unit area of affected peripheral soil” (VDWUA); this ratio determines the volume of dripping water added in ecotoxicological tests on plants;
- Annual ratio “rainwater passing through the deposit / gross mass of sediment passed through” (VWPMS); this ratio makes it possible to set the range of volumes to be percolated in the columns;
- Proportion of percolates from deposits in the river (PPR); this ratio makes it possible to calculate the exposure of aquatic organisms;
- Proportion of percolates from the deposit in the water table immediately downstream of the deposit (PPN); this ratio makes it possible to calculate the residual concentrations in the water table below the deposit.
These ratios, essential to know for the analysis phase (conducting tests), are defined on the basis of a simplified water balance of the site, to be carried out during the problem formulation phase, in particular by means of a study hydrogeology of the site.
Values to protect and evaluation parameters: In the absence of precise information on land uses around the deposit, we must consider: the general protection of species likely to be present, and the preservation of practices such as agriculture.
Therefore, the proposed evaluation parameters can be formulated as follows:
− The deposit of sediment must not disrupt the germination and growth of plants on the site, particularly those of agricultural interest;
− It must also not cause short- or long-term effects on aquatic species through runoff;
− Finally, it must not lead to pollution of the alluvial water table, in particular with a view to its noble uses.
Conceptual model for soil deposition scenarios: Figure 6 summarizes the different elements of the conceptual model, as they emerge from the previous; the plants tested are on the one hand a standard plant and on the other hand, a plant that grows on the soil of the site studied. Aquatic invertebrates are represented by a pelagic crustacean (Ceriodaphnia dubia) and phytoplankton by tests on unicellular algae. The details of these choices are justified in the report Ecotoxicological evaluation of contaminated sediments or dredged materials (II - Presentation of test methods).
The parameters measured include survival, growth, and reproduction, as well as genotoxicity (on amphibians). As in the case of the “gravel pit” scenario, the representation of the ecosystem underlying this model is greatly simplified, for the same reasons.
How to choose criteria and methods?
[71] classify sediment quality criteria into two categories: those that make it possible to establish a cause-effect relationship, but do not easily allow mixtures of polluting substances encountered in the environment to be taken into consideration, and those that, on the contrary, make it possible to do so, but remain weak in the domain of the causal relationship. In the first category, they place the quality criteria obtained by sharing balance and using tests on enriched sediments, in the second the co-occurrence approaches are based on field studies. This classification is not just an academic question if we consider the different uses of sediment quality criteria. They can in fact be used to (1) interpret chemical analysis data, (2) identify substances or sectors at risk, (3) identify the need for more detailed studies in certain sectors, (4) carry out an assessment prior to a discharge, (5) establish a link between a source of pollution and the quality of the sediment, (6) establish regulatory thresholds or (7) restoration objectives. We can clearly see that the criteria for which the cause-effect relationship is well established are essential for uses (4) to (7), while the criteria obtained by the co-occurrence approach on field data should not be used. for these types of uses.
Several recent publications raise the idea that convergence between the different approaches is possible, even desirable. [72] was the first to propose a consensual approach for PAHs, which appears to have received the approval of the EPA. He proposes to retain as the consensual value the arithmetic average of the different criteria (TEL, ERL, SLC, EP on the one hand, PEL, ERM, etc. on the other hand). For their part [73], ensure that the establishment of consensual criteria combines the advantages of the two categories mentioned by [71], that is to say, that they would have a more causal basis. as correlative, and at the same time would take mixtures into account. After grouping the existing criteria into two groups, according to their respective definitions (Table 4), they define a consensual criterion per group by calculating the geometric mean.
The predictive nature of TEC and PEC was tested on 347 results combining chemical analyses and bioassays on freshwater sediments [74], according to two methods: on the one hand the capacity of individual TEC and PEC to classify correctly the sediments, on the other hand, the relationship between average quotients calculated using PECs and frequency of appearance of the effects. The agreement between the concentration of substances and toxicity of sediments (% of toxic samples in the concentration ranges, delimited by TEC and PEC) appears satisfactory for 20 of the 28 chemical parameters studied; the agreement would apparently be improved for mercury, naphthalene, dieldrin, etc. if the value of the corresponding PECs had been lower.
When we apply the quotient method to evaluate the predictive capacity of the criteria in the case of a mixture, we also obtain satisfactory results (in particular, 83% of the samples for which the average of the quotients is < 0.5 are non-toxic; 0.5 is proposed as a benchmark value to classify poly-contaminated sediments according to their toxicity). Likewise, the authors find a good correlation between the incidence of toxicity and the average quotients. Even if the theoretical basis justifying a consensual approach seems quite thin, the practical interest is therefore not negligible. However, these consensual criteria are based on a reasoned selection of existing criteria; on the other hand, the use of consensus criteria, including for regulatory purposes, should take into account the fact that their definition includes a certain percentage of type I (TEC) or II (PEC) errors.
A certain number of works on quality criteria, in water or in sediments, address the problem of adjusting “generic” criteria to the specific conditions of a region or site. This involves, for example, taking into account the influence of water hardness on the toxicity of trace elements [75,76], or the influence of sediment particle size on the bioavailability of contaminants [77]. It may also involve considerations of “natural” concentrations of trace elements, deemed to be supported by organisms, and therefore to be distinguished from “anthropogenic” concentrations in the determination of quality criteria; in this case, the notion of added risk is introduced, and the concentration corresponding to the level of risk retained for the criterion considered is added to the regional background value [45].
Conclusions and perspectives
This study marks the initial phase in developing procedures to evaluate ecosystem risk from dredged materials. It involved illustrating the ecological risk assessment process, creating customized methods for sediment assessment, and testing management scenarios. The study also proposed simplified criteria for evaluating dangers. However, while the process is proven applicable, more experiments are needed to fully demonstrate the operational effectiveness of the proposed conceptual models and ecotoxicological tests due to the limited sample size used in the study. Likewise, it is evident that there are areas within the current risk assessment framework that require further attention and refinement. The choice between TEC or PEC inclusion in the risk quotient for simplified evaluation, the need for more robust effect models, especially in scenarios such as soil deposition, and the consideration of overlooked categories like microflora and microfauna all highlight the need for a comprehensive methodological guide. Additionally, there is a clear necessity for improved exposure models and the calculation of transfer or dilution ratios to ensure accurate risk assessments. Addressing these shortcomings can lead to a more thorough and reliable risk assessment process in the future. The conceptual models proposed for these two scenarios are very simplified models, which raises the question of verifying the relevance of these simplifications – for example by including field approaches. The uncertainty regarding the risk determined at the end of the process was also not examined; However, this is a critical point, particularly with regard to risk management downstream of the evaluation process. Finally, to the extent that other management scenarios can be used, other conceptual models may need to be constructed; However, it would first be appropriate to take an inventory of the management methods for the extracted materials.
- Kennish MJ. Ecology of estuaries: anthropogenic effects. CRC Press. 2019; 512.
- Walker D, Baumgartner D, Gerba C, Fitzsimmons K. Surface water pollution. in, Environmental and pollution science. Elsevier. 2019; 261-292.
- El Amraoui M, Salama Y, El Amraoui B, Lazrak N, Mounkad M. Evaluation of the physicochemical and some trace elements of the surface waters of the Moroccan Atlantic estuary: case of the estuary of the river Bouregreg. Carpathian Journal of Earth and Environmental Sciences. 2015; 10(2):189-198.
- Wojciechowska E, Nawrot N, Walkusz-Miotk J, Matej-Łukowicz K, Pazdro K. Heavy metals in sediments of urban streams: Contamination and health risk assessment of influencing factors. Sustainability. 2019; 11(3):563.
- Hoang HG, Lin C, Tran HT, Chiang CF, Bui XT, Cheruiyot NK, Shern CC, Lee CW. Heavy metal contamination trends in surface water and sediments of a river in a highly-industrialized region. Environmental Technology & Innovation. 2020; 20:101043.
- Tuit C, Wait A. A review of marine sediment sampling methods. Environmental Forensics. 2020; 21(3-4): 291-309.
- Nassiri O, Rhoujjati A, Hachimi ELML. Contamination, sources, and environmental risk assessment of heavy metals in water, sediment, and soil around an abandoned Pb mine site in North East Morocco. Environmental Earth Sciences. 2021; 80(3):96.
- Soriano MCH. Environmental Risk Assessment of Soil Contamination. IntechOpen. 2014; 920.
- McGrath JA, Joshua N, Bess AS, Parkerton TF. Review of Polycyclic Aromatic Hydrocarbons (PAHs) Sediment Quality Guidelines for the Protection of Benthic Life. Integr Environ Assess Manag. 2019 Jul;15(4):505-518. doi: 10.1002/ieam.4142. Epub 2019 Jun 22. PMID: 30945428; PMCID: PMC6852300.
- Niu A, Lin C. Managing soils of environmental significance: A critical review. J Hazard Mater. 2021 Sep 5; 417:125990. doi: 10.1016/j.jhazmat.2021.125990. Epub 2021 May 4. PMID: 34229372.
- Balkaya M. Beneficial use of dredged materials in geotechnical engineering. Recycling and reuse approaches for better sustainability. 2019; 21-38.
- Crocetti P, González-Camejo J, Li K, Foglia A, Eusebi AL, Fatone F. An overview of operations and processes for circular management of dredged sediments. Waste Manag. 2022 Jun 1; 146:20-35. doi: 10.1016/j.wasman.2022.04.040. Epub 2022 May 8. PMID: 35545007.
- Brusseau ML, Pepper I, Gerba C. Environmental and Pollution Science. Elsevier Science. 2019; 656.
- Linthurst RA, Mulkey LA, Slimak MW, Veith GD, Levinson BM Ecological research in the Office of research and development at the US environmental protection agency: an overview of new directions. Environmental Toxicology and Chemistry: An International Journal. 2000; 19(4):1222-1229.
- Kumar V, Sharma A, Pandita S, Bhardwaj R, Thukral AK, Cerda A. A review of ecological risk assessment and associated health risks with heavy metals in sediment from India. International Journal of Sediment Research. 2020; 35(5):516-526.
- Chormare R, Kumar MA. Environmental health and risk assessment metrics with special mention to biotransfer, bioaccumulation and biomagnification of environmental pollutants. Chemosphere. 2022 Sep; 302:134836. doi: 10.1016/j.chemosphere.2022.134836. Epub 2022 May 4. PMID: 35525441.
- Lécrivain N, Aurenche V, Cottin N, Frossard V, Clément B. Multi-contamination (heavy metals, polychlorinated biphenyls and polycyclic aromatic hydrocarbons) of littoral sediments and the associated ecological risk assessment in a large lake in France (Lake Bourget). Sci Total Environ. 2018 Apr 1;619-620:854-865. doi: 10.1016/j.scitotenv.2017.11.151. Epub 2017 Nov 29. PMID: 29734631.
- Gerba CP. Risk assessment. in, Environmental and pollution science. Elsevier. 2019; 541-563.
- Senese V, Boriani E, Baderna D, Mariani A, Lodi M, Finizio A, Testa S, Benfenati E. Assessing the environmental risks associated with contaminated sites: Definition of an Ecotoxicological Classification index for landfill areas (ECRIS). Chemosphere. 2010 Jun;80(1):60-6. doi: 10.1016/j.chemosphere.2010.03.035. Epub 2010 Apr 27. PMID: 20427072.
- Suter GW, Efroymson RA, Sample BE, Jones DS. Ecological Risk Assessment for Contaminated Sites. CRC Press. 2000; 460.
- Asante-Duah K. Hazardous waste risk assessment. CRC Press. 2021; 400.
- Dalezios NR. Environmental Hazards Methodologies for Risk Assessment and Management. IWA Publishing. 2017; 560.
- Suter GW. Introduction to ecological risk assessment for aquatic toxic effects. in, Fundamentals of Aquatic Toxicology. CRC Press. 2020; 803-816.
- Suter II GW. Fundamentals of Environmental Assessment. CRC Press. 2023; 322.
- McBean EA. Risk Assessment: Procedures and Protocols. Wiley. 2018; 336.
- Zhang X, Du H, Wang Y, Chen Y, Ma L, Dong T. Watershed landscape ecological risk assessment and landscape pattern optimization : Take Fujiang River Basin as an example. Human and Ecological Risk Assessment: An International Journal. 2021; 27(9-10):2254-2276.
- Robson MG, Toscano WA, Meng Q, Kaden DA. Risk Assessment for Environmental Health. CRC Press. 2022; 400.
- Singh A, Basak P. Economic and environmental evaluation of municipal solid waste management system using industrial ecology approach: Evidence from India. Journal of Cleaner Production. 2018; 195:10-20.
- Padulosi S, Martelli F, Sciotti A, Putzu D, Filippone M, Mininni G. Environmental risk assessment of conditioned soil: Some Italian case studies. in, Tunnels and Underground Cities. Engineering and Innovation Meet Archaeology, Architecture and Art. CRC Press. 2019; 505-514.
- Rathoure AK. Handbook of Research on Waste Diversion and Minimization Technologies for the Industrial Sector. IGI Global. 2021; 455.
- Khan R, Hossain S, Anik AH, Phoungthong K, Islam ARMT, Saha N, Idris AM, Alam M. Indexical and statistical approaches to investigate the integrated origins of elements in the sediment of Teesta River, Bangladesh: sediment quality and ecological risk assessment. Environ Sci Process Impacts. 2023 Apr 26;25(4):832-849. doi: 10.1039/d2em00475e. PMID: 36897614.
- Muniz DH, Oliveira-Filho EC. Multivariate Statistical Analysis for Water Quality Assessment: A Review of Research Published between 2001 and 2020. Hydrology. 2023; 10(10):196.
- Mudroch A. Manual of Physico-Chemical Analysis of Aquatic Sediments. CRC Press. 2017; 320.
- La Point TW, Fairchild JF. Evaluation of sediment contaminant toxicity: the use of freshwater community structure. in, Sediment toxicity assessment. CRC Press. 2018; 87-110.
- Giesy JP, Hoke RA. Freshwater sediment quality criteria: toxicity bioassessment. in, Sediments. CRC Press. 2020; 265-348.
- Wang W, Gorsuch JW, Hughes JS. Plants for Environmental Studies. CRC Press. 2020; 560.
- Garcia-Reyero N, Murphy CA. A Systems Biology Approach to Advancing Adverse Outcome Pathways for Risk Assessment. Springer International Publishing. 2018; 422.
- Väänänen K, Leppänen MT, Chen X, Akkanen J. Metal bioavailability in ecological risk assessment of freshwater ecosystems: From science to environmental management. Ecotoxicol Environ Saf. 2018 Jan; 147:430-446. doi: 10.1016/j.ecoenv.2017.08.064. Epub 2017 Sep 14. PMID: 28888793.
- Matsuda H. Ecological Risk Management: For Conservation Biology and Ecotoxicology. Springer Nature Singapore. 2021; 314.
- Ishikawa Y, Murata M, Kawaguchi T. Globally applicable water quality simulation model for river basin chemical risk assessment. Journal of Cleaner Production. 2019; 239:118027.
- Wan Mohtar WHM, Abdul Maulud KN, Muhammad NS, Sharil S, Yaseen ZM. Spatial and temporal risk quotient based river assessment for water resources management. Environ Pollut. 2019 May; 248:133-144. doi: 10.1016/j.envpol.2019.02.011. Epub 2019 Feb 8. PMID: 30784832.
- Yang Y, Chen Z, Zhang J, Wu S, Yang L, Chen L, Shao Y. The challenge of micropollutants in surface water of the Yangtze River. Sci Total Environ. 2021 Aug 1; 780:146537. doi: 10.1016/j.scitotenv.2021.146537. Epub 2021 Mar 18. PMID: 33774309.
- Wang N, Wang A, Kong L, He M. Calculation and application of Sb toxicity coefficient for potential ecological risk assessment. Sci Total Environ. 2018 Jan 1;610-611:167-174. doi: 10.1016/j.scitotenv.2017.07.268. Epub 2017 Aug 10. PMID: 28803194.
- Adam V, Yang T, Nowack B. Toward an ecotoxicological risk assessment of microplastics: Comparison of available hazard and exposure data in freshwaters. Environ Toxicol Chem. 2019 Feb;38(2):436-447. doi: 10.1002/etc.4323. Epub 2019 Jan 11. PMID: 30488983; PMCID: PMC6849787.
- Nys C, Van Regenmortel T, Janssen CR, Oorts K, Smolders E, De Schamphelaere KAC. A framework for ecological risk assessment of metal mixtures in aquatic systems. Environ Toxicol Chem. 2018 Mar;37(3):623-642. doi: 10.1002/etc.4039. Epub 2018 Feb 15. PMID: 29135043.
- Svensson N, Norén A, Modin O, Karlfeldt Fedje K, Rauch S, Strömvall AM, Andersson-Sköld Y. Integrated cost and environmental impact assessment of management options for dredged sediment. Waste Manag. 2022 Feb 1; 138:30-40. doi: 10.1016/j.wasman.2021.11.031. Epub 2021 Nov 27. PMID: 34847467.
- Munns WR Jr, Berry WJ, Dewitt TH. Toxicity testing, risk assessment, and options for dredged material management. Mar Pollut Bull. 2002 Apr;44(4):294-302. doi: 10.1016/s0025-326x(01)00250-8. PMID: 12139319.
- Farnelli GM, Tanzi A. Convention on the prevention of marine pollution by dumping of wastes and other matter 1972 and 1996 protocol. in, Elgar Encyclopedia of Environmental Law. Edward Elgar Publishing. 2017; 175-183.
- Lonsdale JA, Blake S, Griffith A. A novel systematic, risk based approach to support the designation of aquatic disposal sites. Mar Pollut Bull. 2021 Jan; 162:111874. doi: 10.1016/j.marpolbul.2020.111874. Epub 2020 Dec 10. PMID: 33310542.
- Solanki P, Jain B, Hu X, Sancheti G. A Review of Beneficial Use and Management of Dredged Material. Waste. 2023; 1(3):815-840.
- Gorton III WT. Continued Turbulence in' Waters of the United States'. Ky. J. Equine Agric. & Nat. Resources L. 2017; 10:237.
- Ennahal I. Valorisation des sédiments de dragage dans des matrices polymères. Ecole nationale supérieure Mines-Télécom Lille Douai. 2019; 224.
- Patmont C, LaRosa P, Narayanan R, Forrest C. Environmental dredging residual generation and management. Integr Environ Assess Manag. 2018 May;14(3):335-343. doi: 10.1002/ieam.4032. Epub 2018 Mar 14. PMID: 29400910.
- Bourmatte N. Granulats recyclés de substitution pour bétons hydrauliques. Université Frères Mentouri-Constantine 1. 2017; 176.
- Lasier PJ, Winger PV, Bogenrieder KJ. Toxicity of manganese to Ceriodaphnia dubia and Hyalella azteca. Arch Environ Contam Toxicol. 2000 Apr;38(3):298-304. doi: 10.1007/s002449910039. PMID: 10667926.
- Huang CW, Yen PL, How CM, Chai ZY, Liao VH. Levels of bioavailable manganese in river sediment may elevate reproductive risk in model organism Caenorhabditis elegans. Aquat Toxicol. 2021 Oct; 239:105958. doi: 10.1016/j.aquatox.2021.105958. Epub 2021 Sep 4. PMID: 34509924.
- Libralato G, Minetto D, Lofrano G, Guida M, Carotenuto M, Aliberti F, Conte B, Notarnicola M. Toxicity assessment within the application of in situ contaminated sediment remediation technologies: A review. Sci Total Environ. 2018 Apr 15; 621:85-94. doi: 10.1016/j.scitotenv.2017.11.229. Epub 2017 Nov 23. PMID: 29179080.
- Ustaoğlu F, Islam MS. Potential toxic elements in sediment of some rivers at Giresun, Northeast Turkey: A preliminary assessment for ecotoxicological status and health risk. Ecological indicators. 2020; 113:106237.
- Cui X, Geng Y, Sun R, Xie M, Feng X, Li X, Cui Z. Distribution, speciation, and ecological risk assessment of heavy metals in Jinan Iron & Steel Group soils from China. Journal of Cleaner Production. 2021 ; 295:126504.
- Gujre N, Mitra S, Soni A, Agnihotri R, Rangan L, Rene ER, Sharma MP. Speciation, contamination, ecological and human health risks assessment of heavy metals in soils dumped with municipal solid wastes. Chemosphere. 2021 Jan; 262:128013. doi: 10.1016/j.chemosphere.2020.128013. Epub 2020 Aug 18. PMID: 33182094.
- Bianco F, Race M, Papirio S, Esposito G. Removal of polycyclic aromatic hydrocarbons during anaerobic biostimulation of marine sediments. Sci Total Environ. 2020 Mar 20; 709:136141. doi: 10.1016/j.scitotenv.2019.136141. Epub 2019 Dec 18. PMID: 31887522.
- Dhar K, Subashchandrabose SR, Venkateswarlu K, Krishnan K, Megharaj M. Anaerobic Microbial Degradation of Polycyclic Aromatic Hydrocarbons: A Comprehensive Review. Rev Environ Contam Toxicol. 2020;251:25-108. doi: 10.1007/398_2019_29. PMID: 31011832.
- Kumar M, Bolan NS, Hoang SA, Sawarkar AD, Jasemizad T, Gao B, Keerthanan S, Padhye LP, Singh L, Kumar S, Vithanage M, Li Y, Zhang M, Kirkham MB, Vinu A, Rinklebe J. Remediation of soils and sediments polluted with polycyclic aromatic hydrocarbons: To immobilize, mobilize, or degrade? J Hazard Mater. 2021 Oct 15;420:126534. doi: 10.1016/j.jhazmat.2021.126534. Epub 2021 Jul 2. PMID: 34280720.
- Marquès M, Cervelló D, Mari M, Sierra J, Schuhmacher M, Domingo JL, Nadal M. The role of iron oxide on the photodegradation of polycyclic aromatic hydrocarbons: Characterization and toxicity. Polycyclic Aromatic Compounds. 2020; 40(2):524-534.
- Ben Othman H, Pick FR, Sakka Hlaili A, Leboulanger C. Effects of polycyclic aromatic hydrocarbons on marine and freshwater microalgae - A review. J Hazard Mater. 2023 Jan 5; 441:129869. doi: 10.1016/j.jhazmat.2022.129869. Epub 2022 Aug 30. PMID: 36063709.
- Rickard D, Mussmann M, Steadman JA. Sedimentary sulfides. Elements. 2017; 13(2):117-122.
- Kolarova N, Napiórkowski P. Trace elements in aquatic environment. Origin, distribution, assessment, and toxicity effect for the aquatic biota. Ecohydrology & Hydrobiology. 2021; 21(4):655-668.
- Hilario Garcia AL, Matzenbacher CA, Santos MS, Prado L, Picada JN, Premoli SM, Corrêa DS, Niekraszewicz L, Dias JF, Grivicich I, da Silva J. Genotoxicity induced by water and sediment samples from a river under the influence of brewery effluent. Chemosphere. 2017 Feb; 169:239-248. doi: 10.1016/j.chemosphere.2016.11.081. Epub 2016 Nov 20. PMID: 27880922.
- Sujitha SB, Lopez-Hernandez JF, García-Alamilla P, Morales-García SS, Márquez-Rocha FJ. Evaluation of polycyclic aromatic hydrocarbons in sediments of Balsas River Mouth, Pacific Coast, Mexico: Sources, risks, and genotoxicity. Chemosphere. 2023 Aug; 332:138898. doi: 10.1016/j.chemosphere.2023.138898. Epub 2023 May 9. PMID: 37169094.
- Williams RJ, Holladay SD, Williams SM, Gogal RM Jr. Environmental Lead and Wild Birds: A Review. Rev Environ Contam Toxicol. 2018; 245:157-180. doi: 10.1007/398_2017_9. PMID: 29038944.
- Destefani A, Friedrichsen JC, Resgalla Jr C. Evaluation of potential confounding factors in sediment toxicity tests with Hyalella azteca (Saussure, 1858). Latin American journal of aquatic research. 2018; 46(4):791-798.
- Zhang Y, Yin J, Qv Z, Chen H, Li H, Zhang Y, Zhu L. Deriving freshwater sediment quality guidelines of polycyclic aromatic hydrocarbons using method of species sensitivity distribution and application for risk assessment. Water Res. 2022 Oct 15; 225:119139. doi: 10.1016/j.watres.2022.119139. Epub 2022 Sep 20. PMID: 36155002.
- Paschal OI, Enyohwo DK. Polychlorinated biphenyls (PCBs) in water and sediments from the Udu River, Niger Delta, Nigeria: concentration, distribution and risk assessment. Journal of Environmental Exposure Assessment. 2022; 1(4):20.
- MacDonald DD, Ingersoll CG, Berger TA. Development and evaluation of consensus-based sediment quality guidelines for freshwater ecosystems. Arch Environ Contam Toxicol. 2000 Jul;39(1):20-31. doi: 10.1007/s002440010075. PMID: 10790498.
- Baran A, Mierzwa-Hersztek M, Gondek K, Tarnawski M, Szara M, Gorczyca O, Koniarz T. The influence of the quantity and quality of sediment organic matter on the potential mobility and toxicity of trace elements in bottom sediment. Environ Geochem Health. 2019 Dec;41(6):2893-2910. doi: 10.1007/s10653-019-00359-7. Epub 2019 Jun 24. PMID: 31236855; PMCID: PMC6856041.
- Li H, Li Y, Guo G, Li Y, Zhang R, Feng C, Zhang Y. Distribution, Site-Specific Water Quality Criteria, and Ecological Risk Assessment of Heavy Metals in Surface Water in Fen River, China. Toxics. 2023 Aug 15;11(8):704. doi: 10.3390/toxics11080704. PMID: 37624209; PMCID: PMC10458924.
- Xu J, Bland GD, Gu Y, Ziaei H, Xiao X, Deonarine A, Reible D, Bireta P, Hoelen TP, Lowry GV. Impacts of Sediment Particle Grain Size and Mercury Speciation on Mercury Bioavailability Potential. Environ Sci Technol. 2021 Sep 21;55(18):12393-12402. doi: 10.1021/acs.est.1c03572. Epub 2021 Sep 10. PMID: 34505768.
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.



 Save to Mendeley
Save to Mendeley
