Open Journal of Environmental Biology
Effects of precipitation and temperature on the species composition and pollinator efficiency of ocimum kilimandscharicum flower visitors in Kakamega forest ecosystem
1Department of Biological Sciences, Masinde Muliro University of Science and Technology, Box 190-50100, Kakamega, Kenya
2National Environmental Management Authority, Box 499-50100, Kakamega, Kenya
Author and article information
Cite this as
HM Tsingalia, Mandela HK (2023) Effects of precipitation and temperature on the species composition and pollinator efficiency of ocimum kilimandscharicum flower visitors in Kakamega forest ecosystem. Open J Environ Biol. 2023; 8(1): 39-47. Available from: 10.17352/ojeb.000038
Copyright License
© 2023 HM Tsingalia, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Pollination, a critical ecosystem service in the maintenance of biodiversity is on the decline due to several factors including habitat loss, exotic pest invasions, pollution, overharvesting, climate, and land use changes. This study analyzed flower visitors’ activity of Ocimum kilimandscharicum in the Kakamega forest. Specifically, the study sought to: (i) assess the effects of temperature and precipitation on flower visitors’ diversity and (ii) identify the most efficient flower visitor using seed set analysis. Data on pollinators were collected through direct observation and sweep-netting and the bagging method in which, flowers were covered using a pollinator bag pre-anthesis and allowed a single visit from a flower visitor. Seed sets from the flowers were collected and counted. Six study sites were identified along two transects each 2.5 km long and labeled A to F. Sampling was done from 7:30 am to 4:00 pm, three days a week for five months consecutively. Secondary data on bee species and their characteristics were used in identification. There were no significant correlations between temperature and diversity (r = -0.509, p = 0.3810), precipitation and diversity (r = 0.377; p = 0.531), temperature and species abundance (r = -0.00618; p = 0.9921), species abundance and precipitation (r = -0.248; p = 0.688), temperature and the species richness of flower-visiting insects (r = -0.729 p = 0.1623) and between precipitation and species richness (r = 0.824; p = 0.08592). The highest number of seed sets, 12,944 was collected under the Apis mellifera making this species, the most efficient pollinator. This study clearly shows that Ocimum kilimandscharicum flower visitors are important in pollination where the higher number of visits translates into higher numbers of seeds set.
Some three-quarters of flowering plants globally and about 35 percent of the world’s food crops depend on animal pollinators to reproduce. Insect pollinators such as butterflies, beetles, and bees have different adaptations that enhance the collection of pollen grains [1-3]. The extent of their activities is determined by the prevailing environmental conditions [4,5]. Insect pollinators play important roles in the maintenance of plant populations, human agricultural enterprise, and commercial value. Pollination and seed dispersal are the most threatened processes of plant regeneration [6]. Consequently, there is an urgent need to evaluate their complex responses to ongoing global changes with respect to their diversity, distribution, abundance, and the services they offer to the ecosystems, particularly in the tropics [7,8]. Flower visitors do not primarily visit to pollinate flowers [9-11]. Some visit the plant to obtain nutritional resources such as water, and nectar, while others seek shade [1,12] and in the process, may cause incidental pollination.
Today, the decline in pollinator distribution and abundance impacts the effective pollination of crops resulting from a multitude of factors that include loss of pollinator habitats due to anthropogenic human activities such as agriculture and urbanization [13]. The widespread application of pesticides has also been linked to the rapid decline in pollinator abundance through their effect on pollinator behaviour, navigation abilities, and success in reproduction. Climate changes through changes in temperature, precipitation patterns, and extreme weather events, are also believed to impact the timing of the emergence of pollinator and migration, as well as food resource availability which further exacerbates decline in pollinator abundance. The deliberate and indeliberate introduction and spread of invasive pathogens and parasites are also known to negatively affect pollinator abundance. Inadequate pollen quality and quantity can also contribute negatively to inadequate crop pollination, which may lead to lower yields [14].
This study evaluated the effects of precipitation and temperature on Ocimum kilimandscharicum flower visitor diversity and abundance in a bid to identify the most efficient flower visitors and their effects on the number of seeds set. Given the limited knowledge of Ocimum kilimandscharicum pollinators, this study sought to determine their significance and value in supporting and enhancing pollination services in forest-adjacent communities. This information is important for pollination awareness among the local communities and ultimately can be used to increase crop yields [15,16] (Navarro, et al. 2003). Furthermore, pollinator knowledge will contribute to evaluating the possibility of pollination of more plant species along forest edges and this will be useful in forestry and the production of commercial crops [17,18] (Wright, 1953). Such knowledge will raise awareness of the importance of pollinators in agroecosystems and consequently, food security [19-22].
In the Kakamega forest ecosystem, Ocimum kilimandscharicum (Basil) is a major plant for insect foraging [23-26] and it is now being cultivated on individual farmlands for commercial purposes among the forest-adjacent communities. It is a wild shrub [27] that grows in the forest and the effects of its domestication are yet to be understood. In addition, the current climate change [28-30] in which a rise in temperature is a major indicator of this crisis may induce different responses in plant and their pollinators [31-33]. Ocimum insect visitors and pollinators are responsive to the prevailing temperature changes [24-36]. While tropical insects are relatively sensitive to temperature changes [37-39]. Deutsch, et al. [24] and Souza, et al. [12] point out that, insect species at higher latitudes have broader thermal tolerance; and any increase in temperature may enhance the performance of insects at higher latitudes [40,41].
Prevailing environmental factors tend to critically influence the relationship between plants and their pollinators. For instance, seasonal variations play crucial roles by acting as drivers of environmental conditions (Memmott, et al. 2007). In addition, local environmental conditions that occur within seasons also affect plants and their pollinators. Rainfall acting as an environmental effect has the potential to impact plant-pollinator interactions because, rain tends to have direct physical effects on both flowers and their pollinators, but also interferes with the timing of pollinator visits [6]. Local temperatures can affect Ocimum pollinator behaviour, altering the number of visits by a single pollinator and pollinators’ behavior within Ocimum flowers [42-44]. On a larger scale, changes in temperature over the entire season may alter the abundance and diversity of pollinators [45]. For instance, pollinators with a narrow temperature tolerance may be replaced by pollinators that are less sensitive to temperature changes or have higher optimal temperatures [40,46,] (Soroye, et al. 2020). Soil enhancement has been shown to significantly influence pollinators by significantly increasing pollinator visit durations and reducing the time to first discovery of flowers [47]. Many flowers are pollinated by the currents of wind or water that act as vectors. Wind pollinator flowers tend to be small with no petals, no special colours, no odours, or nectar. These plants produce enormous numbers of small pollen grains [48]. Some plants have varying pollination methods, including both biotic and abiotic pollination. For instance, the orchid Oeceoclades maculata uses both rain and butterflies, depending on its environmental conditions [49].
Biotic factors can also affect Ocimum flower visitors. These include competition, predators, parasites, and pathogens [28,50]. Competition for resources can play an important role in some insect pollinator declines and consequently, shape the effects of environmental change on pollination services. Competition may vary with distance from the forest as a function of the availability of resources [51,52]. Predators can influence pollinator abundance and foraging behavior. In many cases, predators cause pollinators to visit plants less frequently and for shorter durations [53]. This decline in visitation can lead to pollen limitation and decreased seed set.
Parasites can weaken plant-pollinator mutualistic interactions, as they reduce mutual benefits for one or both partners and can lead to conditional interactions in which outcomes depend on ecological conditions (Bronstein, 1994; Stanton, 2003). Flower sharing amongst pollinator species represents a route for interspecific insect pathogen transmission. Multiple global change pressures can cause plant–pollinator extinctions and this may alter the risks of pathogen-host shifts, intra- or interspecific pathogen spread, and the emergence of novel population or community epidemics. Flowers are hubs for pathogen transmission [54]. Consequently, the structure of plant-pollinator interaction networks may be pivotal in pathogen-host shifts and modulating disease dynamics. Studies conducted by Le Conte & Navajas [55], Graystock, et al. [56], and Huiru et al. [57] showed that natural movements of pollinator species and exchanges of domesticated bees among beekeepers bring them into contact with new pathogens. This may vary with distance from the forest pointing to the importance of conserving the genetic variability among and within important pollinator species to decrease disease-mediated mortality [55,58,59] and maintain the diversity of insect visitors [60,61].
Study methods
Study area
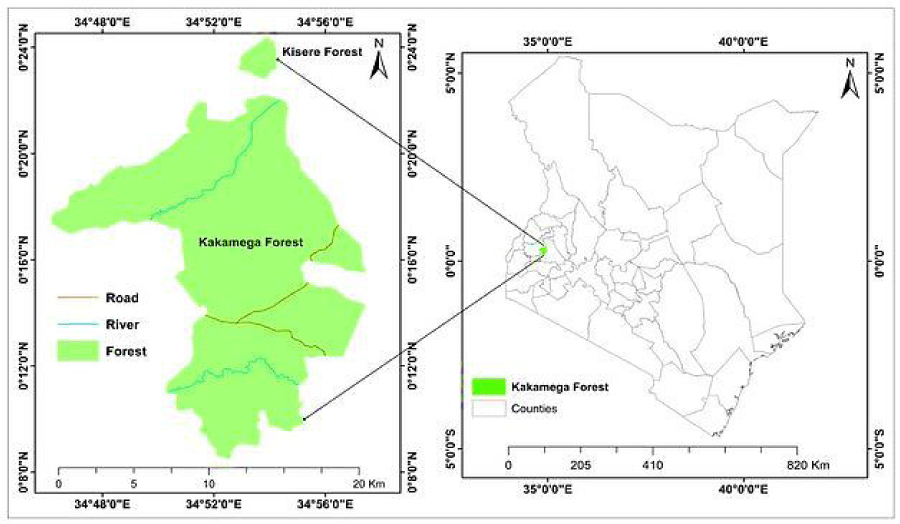
Kakamega forest is a tropical rainforest situated in Western Kenya (Figure 1). Its elevation is about 1500 to 1600 m above sea level with an estimated area of about 240 km2 [62]. Kakamega forest is facing major anthropogenic threats because of the overdependence on it by the local population [63].
The population growth has increased from 406.4 persons per km2 in 2009 to 580 persons per km2 in 2019 [64]. Kakamega Forest receives an average rainfall of 1200mm–1700mm annually that is bimodally distributed. Heavy rainfall is experienced between April and May; it gets slightly dry in June and picks up with the short rains between August and September. It’s usually dry between December and February (Kalinganire, et al. 2001). It is located between latitudes 00˚ 08´ 30.5´ N and 00˚ 22´ 12.5´ N and longitude 34˚ 46´08.0´ E and 34˚ 57´ 26.5´ E. Temperature is fairly constant throughout the year, ranging between 20 ˚C – 30 ˚C (Steinbrecher, 2004). Due to its rich biodiversity and other important ecosystem services Kakamega forest has been ranked as the most sensitive forest ecosystem in Kenya by the International Union of Conservation of Nature (IUCN 1995; Dauber, et al. 2003) [62]. The flora and fauna diversity includes 380 plant species, 350 bird species, 400 butterfly species, and primates [24,62,66].
Transect establishment
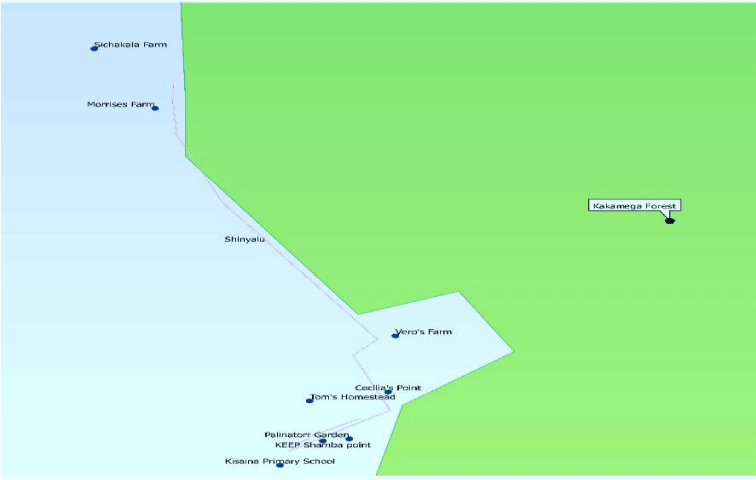
The focus of the study was on flower visitors of Ocimum kilimandscharicum. The main mode of data collection was observation and experimental where flower visitors were captured using a sweep net and control measures were applied to identify the most efficient flower visitor. Two transects of 2.5 km in length, each were initially randomly established running along the forest edge and farmlands cutting through Ocimum kilimandscharicum growing sites [67]. Ocimum kilimandscharicum plots, 4m x 4m with rich floral patches of either naturally growing or cultivated were established along the mapped transects. Transect establishment was based on Ocimum kilimandscharicum availability and where there was least disturbance from livestock grazing and humans. The general surroundings of the quadrats were noted and considered for any possible external influences.
GPS coordinates of these plots were taken and labeled A to F. Sampling sites were located on these transects as demonstrated in Figure 2. These selected sites were all sampled in a similar order in 5 months of collection period for uniformity. Sampling was done for a week in each month and to adequately sample the flower visitors with different diurnal patterns sampling was done within three observation times per day: 7.30 - 11.00 am for morning, 12 noon –1 pm, and 2 – 4 pm for afternoon.
Analysis of the effects of weather parameters on Ocimum kilimandscharicum flower visitors’ diversity and abundance
Temperature and precipitation were taken as the weather parameters that were most likely to affect insect activity [68] in the Kakamega forest. Sampling of flower visitors using a sweep net was done along established transects in the sites with the help of field assistants. Other conditions noted included wind strength and cloud cover of that particular day and described as windy-slightly windy-calm, hot-warn-cold, drizzle-no rain-raining, and clear sky-cloudy-partially cloudy. Temperature and precipitation were taken as a mean on a monthly basis and were obtained from the Kenya Meteorological Department Station in Kakamega.
Determination of the most efficient flower visitor of the Ocimum kilimandscharicum
To effectively determine the most efficient flower visitor of the Ocimum kilimandscharicum, the bagging method [69,70] was applied and seed set in terms of number was used to measure the efficiency of pollinators. To accomplish this, three sites were randomly selected along the already established line transects. This was achieved by clustering the already selected sites into three groups and randomly selecting one site in each cluster as a sampling point.
In each of the three sites, a simple random sampling method was used to select a total of 120 Ocimum kilimandscharicum flower stalks prior to anthesis [71]. The selected flowers were then divided into three parts; those that were left un-interfered with were labeled NP (not protected), those that were protected using pollinator bags and left to receive one insect visitor POV (protected one visit) and finally those that were protected and received no insect visitors PNV (protected no visit) [72]. The main focus was on the POV samples. Once the Ocimum kilimandscharicum flowers opened, the pollination bags were removed and observation of the first flower visitor was made and recorded [73]. Flower visitors were recorded and the flower was re-bagged again to prevent further visits from other arthropods [74]. At this point the flower visited was tagged using a colored band giving the plant a history with its visiting agent and its efficiency in terms of seed number could easily be compared [75].
The flowers were monitored until they set seeds and analysis of the seed set number was done. At the final stage seeds formed by all the selected Ocimum kilimandscharicum flowers on all sites and on each group respectively were collected separately to avoid mixing of results (Dag, et al. 2013). Seed quality in terms of number was recorded for comparison with other seed sets from the other sites.
Data analysis
Input of data was done using Microsoft Excel [76]. The analysis was performed using R 2.10.0 software [77]. Weather data was obtained from the Kenya Metrological Department (Nairobi, Kenya). The data was in means (per month); precipitation (mm), and temperatures (˚C). A T-test analysis was carried out to identify any significant differences between sites that were closer to the forest edge compared to those that were furthest from the forest edge. To determine the significant differences between sites ANOVA was used to compare the diversity and abundance of the flower visitors [78]. Pearson Correlation was used to determine the strength of the relationship between weather parameters and flower visitors.
Determination of the most efficient flower visitor was done using data that were identified according to the colour tag on each flower stalk attached representing the flower visitor. Referencing was done at NMK to identify the flower visitor down to the species name according to its characteristics. Simple linear regression was used to represent the seed number per group. Further descriptive and inferential analysis of data was carried out and this was summarized as mean flower visitors per site and frequency distribution that were all represented graphically.
Results
Effects of weather parameters on Ocimum kilimandscharicum flower visitors’ diversity and abundance
Analysis of the effects of weather parameters: precipitation and temperature, on the flower visitors’ abundance and diversity revealed that these parameters did not significantly influence flower visitor abundance and diversity (Table 1).
Determination of the most efficient pollinator by seed set analysis
Out of the 120 selected individual flowers at each of the three sites, 95 successfully set seeds on Morris’s farm, 76 successfully set seeds in the Pollinator Garden and 104 set seeds on Vero’s farm. A number of the flowers dried up prematurely (n = 43), or were destroyed by termites (n = 28) while others were destroyed by interference from a tractor (n = 14). A total of 42,499 seeds were collected from all the study farms. Of these 19,436 seeds were collected from flowers that were Protected with One Visit (POV), 103 seeds from flowers that were Protected with No Visit (PNV), and 22,960 seeds were collected from flowers that were Not Protected (NP).
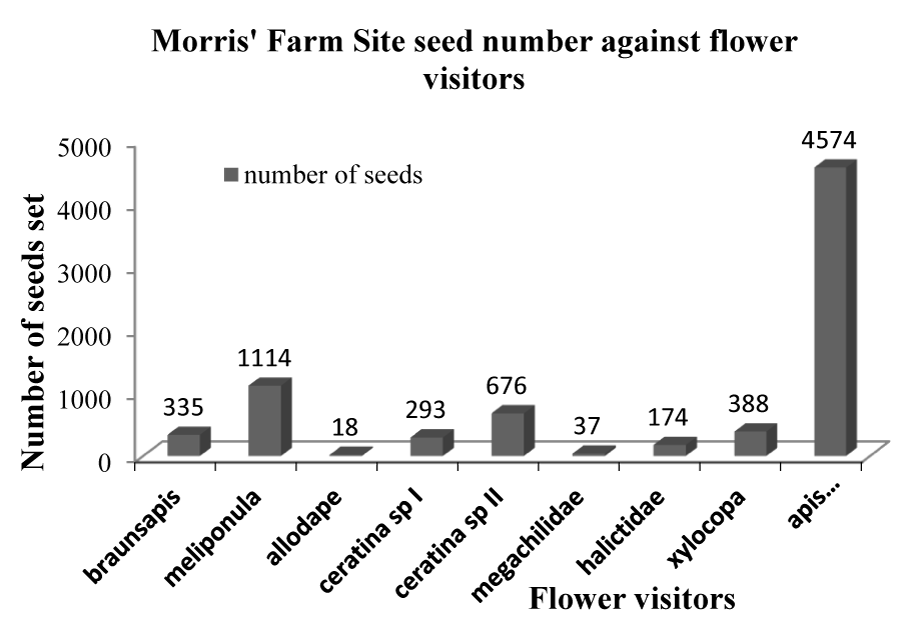
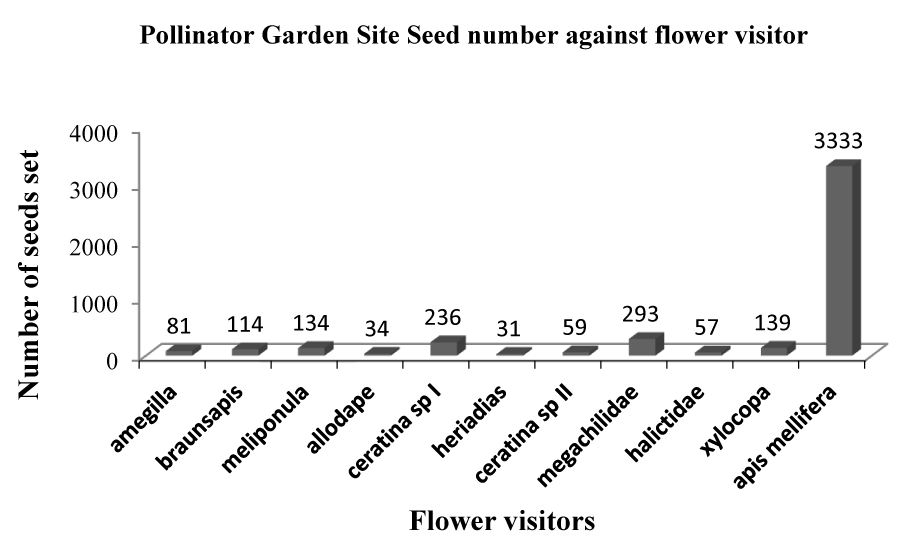
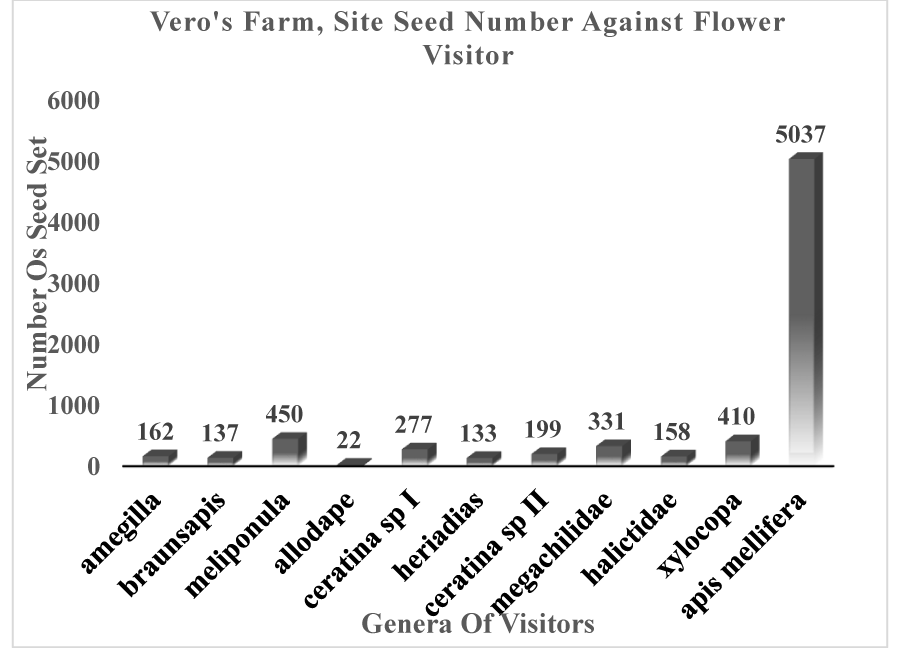
Figures 3-5 clearly show that Apis mellifera visited flowers produced the highest numbers of seeds at 4574, 3333, and 5037 seeds respectively giving a total of 12,944 seeds out of the total 19,436 seeds collected from the POV.
Frequency Visits of individual flower visitors by family/species in selected Sites
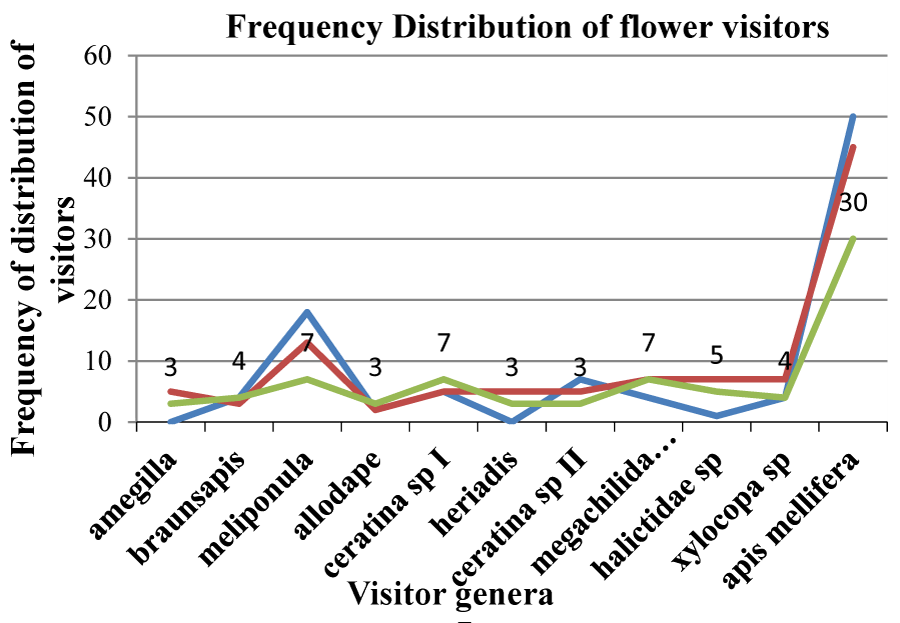
Figure 6 shows the frequency of visits of flower visitors at the three selected study sites; Red for Morris, Yellow for Vero’s, and Blue for the Pollinator Garden.
Results clearly show that the rate at which all flower visitors foraged in the selected Ocimum plant differed. Apis mellifera was the most frequent visitor of flowers in all three sites with a frequency of 30, resulting in the highest number of seed sets produced from Apis mellifera in all the sites. This was followed by Meliponula sp. Ceratina sp. 1 and Megachilidae with a frequency of 7 while the lowest frequency was recorded from Allodape, Amegilla, Ceratina sp. II and Heriadias sp. with a frequency of 3 (Figure 6).
Discussion
This study has clearly demonstrated that Ocimum kilimandscharicum flower visitors play essential roles in pollination. Many of these pollinators are associated with the forest, which is a compelling reason for the conservation of the Kakamega forest. In addition, the study has also shown that the number of visits by flower visitors has implications on the number of seeds realized by the plants. The study also demonstrated that prevailing temperature did not appear to have significant impacts on the flower visitors’ activity, possibly because, these parameters do not show extreme fluctuations in Kakamega forest [79] (Descamps, 2021).
Effects of weather parameters
Temperature did not significantly affect flower visitor diversity and abundance. This is in line with a study by Pereboom & Biesmeijer [80] who reported that despite different bee species facing high solar radiation and high temperatures there seemed to be no major difference in composition between the bees in different biographical areas with different weather patterns. Most bee activity was recorded during the mid-mornings (10 - 11 AM). Pollinators and plants have different climatic requirements, and may therefore respond differently to changes in ambient temperature. Temperature can induce different responses in plants and pollinators. Even if plants and pollinators do respond to the same temperature cue, the strength of the response might differ [31].
The most efficient flower visitor
The large number of seeds (22,960) collected under NP demonstrates effective pollination of the Ocimum kilimandscharicum that occurred with diverse flower visitors. However, under the protected with no visit (PNV), the small number of seeds (103) collected indicates that pollination may have occurred despite the control measures to prevent it. This may have occurred through other forms of pollination, like self-pollination, leaving a knowledge gap that requires further studies.
The complete and actual role of the flower visitors in effective pollination studies is usually dependent on the frequency and relative number of visits to the flowers [81]. According to Bauer & Wing [9], however, results obtained under the single-visit study are equally important in providing an estimate or a foundation for carrying out further studies. It may also indicate a rough estimate of the importance of a flower visitor in pollination and crop production.
Apidae stood out in this study as the most effective pollinators. The study conforms to that of Eeraerts, et al. [82] on sweet cherry where it was found that Apis mellifera were the most efficient pollinator as they helped produce the highest number of seeds in each of the three sites. Morris’ Farm Site was the closest to the forest cover (initially site F which was 50 m from the forest edge); this may also be included as an explanation for the high number of seeds (7609) produced from the site as bees did not have to cover a long distance to get to their foraging sites.
Conclusion
In conclusion, fragmentation, habitat alteration, and disturbances in Kakamega forest will have irreversible effects on the fauna including a wide range of bee flower visitors that play a major role in pollination and are attracted by the Ocimum kilimandscharicum plants. These flower visitors as shown in this study are diverse and are dependent on the forest as they forage along the forest for resources.
Consequently, by planting Ocimum which attracts many diversified pollinators, local communities can benefit from their pollination services to increase their crop yields. While the findings of this study may be inconclusive, because of its short duration, the information obtained forms a foundation and offer baseline data for future studies on pollinators of the Ocimum family.
Recommendation
The diversity and abundance of pollinators observed on Ocimum kilimandscharicum flowers clearly show that this plant species can be used to attract pollinators in the larger farmlands that carry out commercial farming. There is a need for studies to be carried out to identify the significance of Ocimum kilimandscharicum flower visitors in the crop yields of the surrounding communities.
This study also recommends that more detailed studies on the effects of precipitation, temperature, and other weather parameters on Ocimum kilimandscharicum flower visitors with longer study periods in Kakamega forest, be thoroughly investigated in view of the anticipated climate change.
Further recommendations are made for those who domesticate bees for honey production; they can plant Ocimum kilimandscharicum as the study has shown that it is a favorite forage plant among bees including the stingless bees [83].
Finally, there is the need for both researchers and local communities to increase public awareness of Ocimum kilimandscharicum and its preferred flower visitors as this would give more opportunities for further studies to be carried out by researchers.
- Potts SG, Biesmeijer JC, Kremen C, Neumann P, Schweiger O, Kunin WE. Global pollinator declines: trends, impacts and drivers. Trends Ecol Evol. 2010 Jun;25(6):345-53. doi: 10.1016/j.tree.2010.01.007. Epub 2010 Feb 24. PMID: 20188434.
- Armbruster WS. The specialization continuum in pollination systems: diversity of concepts and implications for ecology, evolution, and conservation. Functional Ecology. 2017; 31:88-100.
- Layek U, Das A, Das U. Floral biology, floral volatile organic compounds and floral visitors of Chromolaena odorata, an invasive alien species in West Bengal, India. Biodiversitas. 2022; 23:2118-2129. https://doi.org/10.13057/biodiv/d230447
- Mandela HK, Gikungu M, Lwande W, Tsingalia HM. Distance Effects on Diversity and Abundance of the Flower Visitors of Ocimum Kilimandscharicum in The Kakamega Forest Ecosystem. Accepted Journal of Biodiversity and Environmental Sciences. 2017.
- Lemaitre AB, Pinto CF, Niemeyer HM. Generalized pollination system: Are floral traits adapted to different pollinators? Arthropod-Plant Interactions. 2014; 8:261-272. https://doi.org/10.1007/s11829-014-9308-1
- Neuschulz EL, Mueller T, Schleuning M, Böhning-Gaese K. Pollination and seed dispersal are the most threatened processes of plant regeneration. Sci Rep. 2016 Jul 20;6: 29839. doi: 10.1038/srep29839. PMID: 27435026; PMCID: PMC4951728.
- Martins C, Freitas L. Functional specialization and phenotypic generalization in the pollination system of an epiphytic cactus. Acta Botanica Brasilica. 2018; 32:359-366.
- Rech AR, Jorge LR, Ollerton J, Sazima M. Pollinator availability, mating system and variation in flower morphology in a tropical savanna tree. Acta Botanica Brasilica. 2018; 32:462-472.
- Bauer DM, Wing IS. Economic consequences of pollinator declines: a synthesis. Agricultural and Resource Economics Review. 2010; 39(3): 368-383.
- Quinet M, Jacquemart AL. Troubles in pear pollination: Effects of collection and storage method on pollen viability and fruit production. Acta Oecologica. 2020; 105:103558. https://doi.org/10.1016/j.actao.2020.103558
- da Santos RS, Carneiro LT, de Oliveira Santos JP, da Silva MM, de Oliveira Milfont M, Castro CC. Bee pollination services and the enhancement of fruit yield associated with seed number in self-incompatible tangelos. Scientia Horticulturae. 2021; 276:109743.
- Souza CS, Maruyama PK, Aoki C, Sigrist MR, Raizer J, Gross CL, de Araujo AC. Temporal variation in plant-pollinator networks from seasonal tropical environments: Higher specialization when resources are scarce. Journal of Ecology. 2018; 106:2409-2420.
- F.A.O. Why Bees Matter. 2023. https://www.fao.org/3/I9527EN/i9527en.PDF
- Reis E, Silva-Mann R, Damião M, de Souza E, Franco DF. Pollen Quality and Quantity Effect on Fruit Set and Fruit Quality in Coffee Trees. J. Plant Growth Regul. 2020; 39: 919-928. http://journal.um-surabaya.ac.id/index.php/JKM/article/view/2203.
- Burkle LA, Runyon JB. The smell of environmental change: Using floral scent to explain shifts in pollinator attraction. Appl Plant Sci. 2017 Jun 5;5(6):apps.1600123. doi: 10.3732/apps.1600123. PMID: 28690928; PMCID: PMC5499301.
- Cézar K, Franklin E, Pinto CE. Temporal variation in pollinators’ visitation of Lantana camara in a tropical urban landscape: does butterfly abundance and richness drive the fruit set? Ekológia (Bratislava). 2022; 41:46-56.
- Courtice B, Hoebee SE, Sinclair S, Morgan JW, Courtice B, Hoebee SE, Sinclair S, Morgan JW. Local population density affects pollinator visitation in the endangered grassland daisy Rutidosis leptorhynchoides (Asteraceae). Australian Journal of Botany. 2020; 67:638-648.
- Lemanski NJ, Cook CN, Ozturk C, Smith BH, Pinter-Wollman N. The effect of individual learning on collective foraging in honey bees in differently structured landscapes. Animal Behaviour. 2021; 179:113-123.
- Brosi BJ, Daily GC, Shih TM, Oviedo F, Durán G. The effects of forest fragmentation on bee communities in tropical countryside. Journal of Applied Ecology. 2008; 45, 773-783.
- Fisogni A, Rossi M, Sgolastra F, Bortolotti L, Bogo G, de Manincor N, Quaranta M, Galloni M. Seasonal and annual variations in the pollination efficiency of a pollinator community of Dictamnus albus L. Plant Biol (Stuttg). 2016 May;18(3):445-54. doi: 10.1111/plb.12417. Epub 2015 Dec 7. PMID: 26573095.
- Delle-Vedove R, Schatz B, Dufay M. Understanding intraspecific variation of floral scent in light of evolutionary ecology. Ann Bot. 2017 Jul 1;120(1):1-20. doi: 10.1093/aob/mcx055. PMID: 28873948; PMCID: PMC5737645.
- Nagasaki O. Functional specialization for pollination by scoliid wasps and solitary bees of Ampelopsis glandulosa (Vitaceae). Flora: Morphology, Distribution, Functional Ecology of Plants. 202; 1 284:151921.
- Githinji C, Kokwaro J. Review Plectranthus: Ocimum an Overview of Classification. Journal of Ethnopharmacology. 1995; 103:1-24.
- Gikungu MW. Bee diversity and some aspects of their ecological interactions with plants in a successional tropical community. Doctoral Dissertation, Universitäts-und Landesbibliothek Bonn. 2006.
- Funamoto D. Plant-pollinator interactions in East Asia: a review. Journal of Pollination Ecology. 2019; 25(6): 46-68.
- Jia LB, Huang SQ. An examination of nectar production in 34 species of Dendrobium indicates that deceptive pollination in the orchids is not popular. Journal of Systematics and Evolution. 2022; 60(6): 1371-1377.
- Joshi RK. Chemical Composition of the Essential Oil of Camphor Basil Ocimum kilimandscharicum. Global Journal of Medical Plant Research. 2013; 1 (2):207-209.
- Khojl M, Nielsen A, Stenseth JC. Pollination Services for Sustainable Agriculture; Potential Effects of Climate Change on Crop Pollination. Ecological Letters. 2011; 11:253-244.
- Allen-Perkins A, Magrach A, Dainese M, Garibaldi LA, Kleijn D, Rader R, Reilly JR, Winfree R, Lundin O, McGrady CM, Brittain C, Biddinger DJ, Artz DR, Elle E, Hoffman G, Ellis JD, Daniels J, Gibbs J, et al. CropPol: A dynamic, open and global database on crop pollination. Ecology. 2022 Mar;103(3):e3614. doi: 10.1002/ecy.3614. Epub 2022 Feb 17. PMID: 34921678.
- Biella P, Tommasi N, Guzzetti L, Pioltelli E, Labra M, Galimberti A. City climate and landscape structure shape pollinators, nectar and transported pollen along a gradient of urbanization. Journal of Applied Ecology. 2022; 9:1586-1595.
- Hegland SJ, Nielsen A, Lázaro A, Bjerknes AL, Totland Ø. How does climate warming affect plant-pollinator interactions? Ecol Lett. 2009 Feb;12(2):184-95. doi: 10.1111/j.1461-0248.2008.01269.x. Epub 2008 Nov 26. PMID: 19049509.
- Mason L, Arathi HS. Assessing the efficacy of citizen scientists monitoring native bees in urban areas. Global Ecology and Conservation. 2019; 17: e00561.
- Meiners JM, Griswold TL, Carril OM. Decades of native bee biodiversity surveys at Pinnacles National Park highlight the importance of monitoring natural areas over time. PLoS One. 2019 Jan 17;14(1):e0207566. doi: 10.1371/journal.pone.0207566. PMID: 30653514; PMCID: PMC6336250.
- Deutsch CA, Tewksbury JJ, Huey RB, Sheldon KS, Ghalambor CK, Haak DC, Martin PR. Impacts of climate warming on terrestrial ectotherms across latitude. Proc Natl Acad Sci U S A. 2008 May 6;105(18):6668-72. doi: 10.1073/pnas.0709472105. Epub 2008 May 5. PMID: 18458348; PMCID: PMC2373333.
- Rubene D, Schroeder M, Ranius T. Diversity patterns of wild bees and wasps in managed boreal forests: effects of spatial structure, local habitat, and surrounding landscape. Biological Conservation. 2015; 184:201-208.
- Mola JM, Richardson LL, Spyreas G, Zaya DN, Pearse IS. Long‐term surveys support declines in early-season forest plants used by bumblebees. Journal of Applied Ecology. 2021; 58:1431-1441.
- Steffan-Dewenter I, Potts SG, Packer L. Pollinator diversity and crop pollination services are at risk. Trends Ecol Evol. 2005 Dec;20(12):651-2; author reply 652-3. doi: 10.1016/j.tree.2005.09.004. Epub 2005 Oct 7. PMID: 16701452.
- Steffan-Dewenter I, Westphal C. The Interplay of Pollination Diversity, Pollination Services and Landscape Change. Journal of Applied Ecology. 2009; 45(3):737-741.
- Zhu G, Gutierrez Illan J, Looney C, Crowder DW. Assessing the ecological niche and invasion potential of the Asian giant hornet. Proc Natl Acad Sci U S A. 2020 Oct 6;117(40):24646-24648. doi: 10.1073/pnas.2011441117. Epub 2020 Sep 22. PMID: 32963093; PMCID: PMC7547231.
- Hoyle M, Hayter K, Cresswell JE. Effect of pollinator abundance on self-fertilization and gene flow: application to GM Canola. Ecol Appl. 2007 Oct;17(7):2123-35. doi: 10.1890/06-1972.1. PMID: 17974346.
- Geissler C, Davidson A, Niesenbaum RA. The influence of climate warming on flowering phenology in relation to historical annual and seasonal temperatures and plant functional traits. PeerJ. 2023 Apr 21; 11:e15188. doi: 10.7717/peerj.15188. PMID: 37101791; PMCID: PMC10124540.
- Cruz DDO, Freitas BM, Silva LAD, Silva EMSD, Bomfim IGA. Pollination efficiency of the stingless bee Melipona subnitida on greenhouse sweet pepper. Pesquisa Agropecuária Brasileira. 2005; 40: 1197-1201.
- Petanidou T, Ståhls G, Vujić A, Olesen JM, Rojo S, Thrasyvoulou A, Tscheulin T. Investigating plant-pollinator relationships in the Aegean: The approaches of the project POL-AEGIS (The pollinators of the Aegean archipelago: Diversity and threats). Journal of Apicultural Research. 2013; 52(2): 106-117.
- Petanidou T, Kallimanis AS, Lazarina M, Tscheulin T, Devalez J, Stefanaki A, Hanlidou E, Vujić A, Kaloveloni A, Sgardelis SP. Climate drives plant-pollinator interactions even along small-scale climate gradients: the case of the Aegean. Plant Biol (Stuttg). 2018 Jan;20 Suppl 1:176-183. doi: 10.1111/plb.12593. Epub 2017 Jul 20. PMID: 28637086.
- Kerr JT, Pindar A, Galpern P, Packer L, Potts SG, Roberts SM, Rasmont P, Schweiger O, Colla SR, Richardson LL, Wagner DL, Gall LF, Sikes DS, Pantoja A. CLIMATE CHANGE. Climate change impacts on bumblebees converge across continents. Science. 2015 Jul 10;349(6244):177-80. doi: 10.1126/science.aaa7031. PMID: 26160945.
- Shrestha M, Garcia JE, Bukovac Z, Dorin A, Dyer AG. Pollination in a new climate: Assessing the potential influence of flower temperature variation on insect pollinator behaviour. PLoS One. 2018 Aug 1;13(8):e0200549. doi: 10.1371/journal.pone.0200549. Erratum in: PLoS One. 2018 Aug 23;13(8):e0203153. PMID: 30067757; PMCID: PMC6070230.
- Huang ZY, Giray T. Factors Affecting Pollinators and Pollination. Psyche (Special Issue), 4 pages. 2012. doi:10.1155/2012/302409
- Walker T. Pollination: The Enduring Relationship Between Plant and Pollinator. Princeton University Press. 2020; 1-224. ISBN 9780691203751.
- Armbruster WS . Evolution of Plant-Pollinator Relationships. Cambridge, UK: Cambridge University Press. 2012; 45-67.
- Deguines N, de Flores M, Loïs G, Julliard R, Fontaine C. Fostering close encounters of the entomological kind. Frontiers in Ecology and the Environment. 2018; 16:202-203.
- Ghini R, Morandi MAB. Biotic and abiotic factors associated with soil suppressiveness to Rhizoctonia solani. Scientia Agricola. 2006; 63: 153-160.
- Nielsen A, Reitan T, Rinvoll AW, Brysting AK. Effects of competition and climate on a crop pollinator community. Agriculture, Ecosystems & Environment. 2017; 246: 253-260. https://doi.org/10.1016/j.agee.2017.06.006
- Benoit AD, Kalisz S. Predator Effects on Plant-Pollinator Interactions, Plant Reproduction, Mating Systems, and Evolution. Annual Review of Ecology, Evolution, and Systematics. 2020; 51:1, 319-340.
- Proesmans W, Albrecht M, Gajda A, Neumann P, Paxton RJ, Pioz M, Polzin C, Schweiger O, Settele J, Szentgyörgyi H, Thulke HH, Vanbergen AJ. Pathways for Novel Epidemiology: Plant-Pollinator-Pathogen Networks and Global Change. Trends Ecol Evol. 2021 Jul;36(7):623-636. doi: 10.1016/j.tree.2021.03.006. Epub 2021 Apr 14. PMID: 33865639.
- Le Conte Y, Navajas M. Climate change: impact on honey bee populations and diseases. Rev Sci Tech. 2008 Aug;27(2):485-97, 499-510. English, French. PMID: 18819674.
- Graystock P, Goulson D, Hughes WO. Parasites in bloom: flowers aid dispersal and transmission of pollinator parasites within and between bee species. Proc Biol Sci. 2015 Aug 22;282(1813):20151371. doi: 10.1098/rspb.2015.1371. PMID: 26246556; PMCID: PMC4632632.
- Jia H, Liu Y, Li X, Li H, Pan Y, Hu C, Zhou X, Wyckhuys KAG, Wu K. Windborne migration amplifies insect-mediated pollination services. Elife. 2022 Apr 13; 11:e76230. doi: 10.7554/eLife.76230. PMID: 35416148; PMCID: PMC9042232.
- Aguilar R, Quesada M, Ashworth L, Herrerias-Diego Y, Lobo J. Genetic consequences of habitat fragmentation in plant populations: susceptible signals in plant traits and methodological approaches. Mol Ecol. 2008 Dec;17(24):5177-88. doi: 10.1111/j.1365-294X.2008.03971.x. PMID: 19120995.
- López-Uribe MM, Soro A, Jha S. Conservation genetics of bees: advances in the application of molecular tools to guide bee pollinator conservation. Conservation Genetics. 2017; 18: 501-506.
- Nkoba K, Raina SK, Muli E, Mithöfer K, Mueke J. Species richness and nest dispersion of some tropical meliponine bees (Apidae: Meliponinae) in six habitat types in the Kakamega forest, western Kenya. International Journal of Tropical Insect Science. 2012; 32(4): 194-202.
- de Matos Barbosa M, Jaffé R, Carvalho CS, Lanes ÉC, Alves-Pereira A, Zucchi MI, Alves DA. Landscape influences genetic diversity but does not limit gene flow in a Neotropical pollinator. Apidologie. 2022; 53(4): 48.
- Althof AJ. Human Impact on Flora and Vegetation of Kakamega Forest, Kenya PhD Dissertation. Universität Koblenz, Fachbereich. 2005; 3.
- Vuyiya E, Konje M, Tsingalia H, Obiet L, Kigen C, Wamalwa S, Nyongesa H. Impacts of Human Activities on Tree Species Richness and Diversity in Kakamega Forest. Int. J. Biodiv. & Conserv. 2014; 6(6):428-435.
- Kenya National Bureau of Statistics. Kenya Population and Housing Census Volume II: Distribution of Population by Administrative Units. KNBS. 2019.
- IUCN . Uganda Nationally Threatened Taxa listed at WCMC: conservation listing of plants. The World Conservation Monitoring Centre, Cambridge. 1995.
- Gikungu M, Wittmann D, Irungu D, Kraemer M. Bee diversity along a forest regeneration gradient in Western Kenya. Journal of Apicultural Research. 2011; 50(1): 22-34.
- Winfree R, Williams NM, Gaines H, Ascher JS, Kremen C. Wild bee pollinators provide the majority of crop visitation across land-use gradients in New Jersey and Pennsylvania, USA. Journal of Applied Ecology. 2007; 61(45):794-803.
- Niaz Morshed MD, Abdullah MD, Mamun Al, Sheikh Nihad AI, Mahfuzur Md, Rahman, Naznin Sultana, Mezanur Rahman MD. Effect of weather variables on seasonal abundance of rice insects in southeast coastal region of Bangladesh, Journal of Agriculture and Food Research. ISSN 2666-1543. 2023; 11:100513. https://doi.org/10.1016/j.jafr.2023.100513.
- Szmek KJ, Zych M. Flower Visitor & Pollen Transport Network in a Large City Structure and Properties. Arthropod-Plant interaction. 2013; 7:503-506.
- Razo-León AE, Muñoz-Urias A, Campos-Porras MG, Huerta-Martínez FM, Fierros-López HE. Flower visitors and efficient pollinators of Opuntia joconostle F.A.C.Weber. ex Diguet in Jalisco, Mexico. Bradleya. 2022; (40):19-26.
- Noor S, Tajik O, Golzar J. Simple Random Sampling. International Journal of Education & Language Studies. 2022; 1(2): 78-82. doi: 10.22034/ijels.2022.162982
- Grange MC, Munoz F, Moretti M, Varona-Y-Varona S, Renaud J, Colace MP, Gueguen M, Gallien L. Designing sampling protocols for plant-pollinator interactions - timing, meteorology, flowering variations and failed captures matter. Botany Letters. 2021; 168:3; 324-332, DOI: 10.1080/23818107.2021.1964596
- Delaplane KS, Dag A, Danka RG, Freitas BM, Garibaldi LA, Goodwin MR, Hormaza JL. Standard methods for pollination on research with Apis mellifera. Journal of Apicultural Research. 2013; 52(4):43-51.
- Cecala JM, Lau PW, Leong JM. Floral bagging differentially affects handling behaviours and single‐visit pollen deposition by honey bees and native bees. Eco. l Entomol. 2020:45(5):1099-1107. https://doi.org/10.1111/een.1289
- Vaissiere B, Freitas B, Gemmil-Heren B. Protocol to Detect & Assess Pollination Deficit in Crops: A Handbook for its use. FAO. Rome, Italy. 2011.
- Yee T. The VGAM Package for Categorical Data Analysis. University of Auckland – New Zealand. 2010.
- R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria. 2021.
- Mpondo FT, Ndakidemi PA, Mukma SC, Treydte AC. Insect visitation and pollination networks across traditional rangeland management categories in a Northern Tanzanian rangeland. Global Ecology and Conservation . 2023; 46 : e02581.
- Tsingalia HM. Habitat destruction and severity patterns of abundance in Kakamega Forest, Western Kenya. African Journal of Ecology. 2009; 28:213-226.
- Pereboom JJ, Biesmeijer JC. Thermal constraints for stingless bee foragers: the importance of body size and coloration. Oecologia. 2003 Sep;137(1):42-50. doi: 10.1007/s00442-003-1324-2. Epub 2003 Jun 28. PMID: 12838404.
- Garibaldi LA, Carvalheiro LG, Leonhardt SD, Aizen MA, Blaauw BR, Isaacs R, Winfree R. From research to action: enhancing crop yield through wild pollinators. Frontiers in Ecology and the Environment. 2014; 12(8): 439-447.
- Eeraerts M, Vanderhaegen R, Smagghe G, Meeus I. Pollination efficiency and foraging behaviour of honey bees and non‐Apis bees to sweet cherry. Agricultural and Forest Entomology. 2020; 22(1): 75-82.
- Macharia JK, Raina SK, Muli EM. Stingless Bee Keeping; an Incentive for Rain Forest Conservation in Kenya. Apiacta Journal. 2010; 6:7-15.
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.


 Save to Mendeley
Save to Mendeley
