Open Journal of Environmental Biology
Chelate-assisted phytoextraction using Brassicaceae plants
Biotechnology and Plant Improvement Laboratory, Centre of Biotechnology of Sfax, University of Sfax, P.O. Box 1177, Road Sidi Mansour 6 km, Sfax 3018, Tunisia
Author and article information
Cite this as
Saibi W, Brini F (2020) Chelate-assisted phytoextraction using Brassicaceae plants. Open J Environ Biol. 2020; 5(1): 22-26. Available from: 10.17352/ojeb.000017
Copyright License
© 2020 Saibi W, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.In these last decades, excessive metal concentration pose serious contamination in soils. Therefore, it is urgent to develop and adopt a new strategy and technology to remove soil contaminants. Here, the phytoextraction was considered as a recently developed approach to clean up metal-polluted soils in that the plants are used to translocate the toxic metals from the contaminated soils. Therefore, this mini-review provides a perfection of recent knowledge related to the heavy metal phytoextraction from soils and their accumulation in plants. It has been reported that EDTA enhances the availability of heavy metal to plants and thus increases the accumulation in their shoots. In addition, fundamental and basic advancement has been made on the molecular aspects and the physiological regarding tolerance and metals phytoaccumulation in plants. A multidisciplinary strategy is warranted to make phytoextraction a plausible trade technology to remediate metal-polluted soils. Hence, the purpose is to integrate soil-related and biological progress to suggest research needs and future direction.
The civilization of mankind and the agriculture sustainability rely on two precious bio-resources namely land and water [1,2]. So, the two last ones have been subjected to an hyper exploration and they were severely polluted by kinds of contaminants having its origin from various anthropogenic studies [2,3].
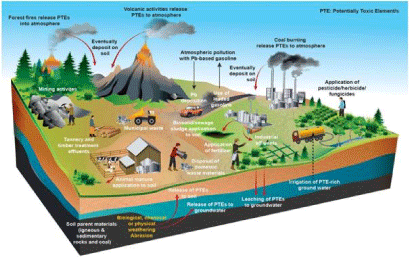
The potentially toxic elements can occur naturally in soils at various amounts depending on the source rock geochemical compositions and soil formation processes like weathering sedimentation and volcanic eruptions. Meanwhile, anthropogenic activities such mining and smelting operations, industrial production, oil and gas production, agricultural activities and military practices can also result in elevated soil contaminant amounts. Indeed Figure 1 illustrates the potentially toxic element sources in soil ecosystems [4,5].
It is crucial to note that the majority of contaminants get accumulated in plants and either directly or indirectly, find their way into food web thus cause severe consequences [3]. In addition, the plants undergo several abiotic stresses such as salt, drought and heavy metal witch are known among the most limiting ones [6-10]. Indeed, it is crucial to signal that as much as one-half of the world irrigated areas are affected by the high salinity level [7,8,10,11], the drought and also the excessive soil metal amount [6,7,12]. The last cited one pose significant hazard to human, plant, animal and health, hence to the ecosystem [7,13,14]. We indicate in this case that soil contamination by toxic metal has often resulted from human activities, such as those related to application of sewage sludge to agricultural soils, industrial emissions, mining, leakage and/ or disposal of industrial wastes and also the pesticide use [12,15]. It is basic also to signal that the phytoextraction is one of the metal phytoremediation technologies as illustrated in Table 1 [16].
Due to the potential toxicity and the high metals persistence, those facts constitute a serious environmental problem that requires an affordable solution [15,17]. Hence, phytoextraction seems to be the most promising technique and has received increasing attention from researchers since it was proposed by various research teams such as [18] as a technology for reclaiming metal polluted soils [3,7,12].
The metal phytoextraction from the soil relies on the use of plants to extract and translocate metals to their harvestable parts [3,16,19,20]. The phytoextraction aim consists on the reduction of the metal concentration in contaminated soils to regulatory levels within a reasonable time frame. This extraction process depends directly on the able duality of the selected plants to grow and accumulate metals under the specific climatic and soil conditions of the site being remediated [16,20].
In another case, it is fundamental to note that the Brassicaceae plant family represents hundreds of plant families reported so far for their potential use in the remediation of varied environmental contaminants including toxic metals and metalloids. The most of the members of the Brassicaceae plant family well represent the metal hyperaccumulation among 0.2% of all angiosperms and thus, have key role in phytoremediation technology. Many of the plant species within Brassicaceae family such as Alyssum, Arabidopsis, Berkheya, Bornmuellera, Cardamine, Cochlearia, Crambe, Peltaria, Stanleya, Thlaspi including oilseed Brassicas grow fast, yield high biomass and are well adapted to a range of environmental conditions [19,20,21].
Metal extraction process
The heavy metal extraction process is due to two approaches that have currently been used to reach this purpose: the first one results in the exploration of plants with exceptional, natural metal-accumulating capacity, so called hyperaccumulators [22], and the second one is the use of high-biomass crop plants with a chemically enhanced phytoextraction method [6,12,21-25]. So, the basic properties of those two phytoextraction strategies of metals from soils are illustrated as followed in Table 2. In addition, the analysis of the Table 2 content and based on some of other’s investigations give birth to a fundamental conclusion summarized on the fact that the natural metal-accumulating capacity seem to be the most one because of the negative repercussion of the chemically enhanced phytoextraction method [26-28].
Metal hyperaccumulation and tolerance in plants
The metal hyperaccumulation capability is a rare phenomenon in plants. Occurring ≈400 vascular plant species in which vast majority of those ones discovered so far was being Ni hyperaccumulators. The plant species having the ability to accumulate Zn, Cd, Pb, Cu and Co are much less numerous [23]. The hyperaccumulation concept has been extended to a plant growing in its natural habitat in that those metal concentrations have been canned in the dry matter of any above ground tissue. This more detailed definition includes plants which accumulate metals in their aerial tissues other than leaves, that might be useful to phytoextraction as well, and disqualify any species that hyperaccumulate metals under synthetic conditions like massive metals addition to the soil or the nutrient solution [13].
The correlation and also the relationship between metal hyperaccumulation and tolerance is still a subject of discussion. Some authors suggest the non-correlation between those traits, while others suggest that hyperaccumulators possess a high degree of metals tolerance. [6] compiled a number of studies in that the metal accumulation meaning tolerant and non-tolerant plants had been compared [29]. Those funding’s led to conclude that there is no pattern regarding accumulation and tolerance [6]. Both shoot and root concentrations are equally variable even when only one particular metal is [7,30,31]. However, at least in some cases, it is clear that the increased tolerance give birth to greater metal amount. Plants may use two strategies to deal with high metal amounts adjacent to their roots: (i) exclusion mechanisms by which the uptake and/or root to shoot transport of metals are restricted; and (2i) internal tolerance mechanisms which immobilize, compartmentalize or detoxify metals in the symplasm through production of metal binding compounds. Given that the phytoextraction purpose is to maximize metal accumulation effeciency in plant tissues, mechanisms of internal tolerance are likely to be crucial and [6,19,32].
Stress-associated protein provides tolerance to heavy metals
The boom in industrialization over the past few decades has led to the onset of long-term pollution by heavy metals as well as serious environmental and also ecological problems for humans, plants and animals. In addition, we signal that some heavy metals have a crucial role during various physiological processes in plants [33,34]; Yet, when present in excessive concentrations in the soil, they can inactive biomolecules, block functional proteins, or displace other essential metal ions and, hence, become toxic causing serious ecological problems. We recently showed that overexpression of LmSAP, a member of the stress-associated protein (SAP) gene family isolated from Lobularia maritima, in transgenic plants led to enhanced tolerance to metal stresses (Cd, Cu, Mn and Zn). Indeed, LmSAP expression increased after 12 h of treatment with those metals, suggesting its involvement in the plant response to heavy metal stress [34]. LmSAP transgenic tobacco plants subjected to these stress conditions were healthy, experienced higher seedling survival rates, and had longer roots than non-transgenic plants. However, they exhibited higher tolerance towards cadmium and manganese than towards copper and zinc [7,30,33,35,36].
Chemical amendments upgrades metal phytoextraction
The solubility and availability of metal are both dependent on the soil characteristics and are strongly influenced by pH [4] and also the level of complexation with soluble ligands [37,38]. So, the metals exist in soil under various organization types: in solution as ionic or organically complexed species; on exchange sites of reactive soil components; complexed with organic matter; occluded in Fe, Al, and Mn oxides and hydroxides; entrapped in primary and secondary minerals [37]. Most metals in soils exist in unavailable forms, thus soil conditions have to be altered to elicit phytoextraction since the phenomenon, depends on a relatively abundant source of soluble metal to enable significant metal uptake and translocation to shoots. In addition, some metals have their extraction rate limited by their inherently low solubility. In such a case, the organic compounds may be used as amendments to upgrade phytoextraction. Such substances can chelate metal ions, therefore modifying the metals availability in soils [4,37,38].
At the same case, we note that as indicated by [39], DTPA and EDTA were noticeably effective in desorbing Pb whereas the natural organic acids solubilized very small amount of this metal from soil. Actually, the lower efficiency of LMWOA in making soil-Pb available to plants compared to synthetic chelates had already been demonstrated. The major drawback for using synthetic chelates (especially EDTA) in field conditions is their high persistence to biodegradation, with consequent high risks of metal leaching through soil profile. The heavy metal levels of in the soil solution after EDTA treatment can be much higher than plant's capabilities to absorb them. Indeed, the analysis of the fundings followed in Table 3 shows that EDTA and DTPA can solubilize up to 77 and 100% of Pb present in the soil samples, respectively. This was equivalent to bring 383 (EDTA) and 521 mg kg-1 (DTPA) of Pb into the soil solution. Of course, such high values must be weighted against the use of these chelates for phytoextraction in the field [6,39].
The chelating agent and its use in Brassicaceae plants for phyto-extraction
Concerning the chelating agent and its use in Brassicaceae plants for phyto-extraction, various studies and investigations were described the effect of the heavy metal and the EDTA application on plant growth and the phyto-extraction potential. Indeed, the pot experiment was conducted to evaluate the phyto-extraction capability of heavy metals by Sorghum. The last one was grown in soil artificially contaminated with a range of heavy metals at various concentrations like lead (300, 350 and 400 mg/kg), chromium (50,100 and 150 mg/kg) and cadmium (100, 150 and 200 mg/kg). In this case, 5 mM EDTA was applied, as chelating agent to the plants after 4 weeks of sowing. Plants were grown for a total of two months and fresh weight and dry weight of shoot and heavy metal accumulation were analyzed at 6 and 8 weeks after sowing. The fundings revealed that the described heavy metals application and EDTA adversely affected shoot length, fresh weight and dry weight of the biological matrix used at both time intervals. Hence, heavy metals uptake increased with the increment of heavy metal by the used plant. The use of 5mM EDTA upgrades the uptake of heavy metal [40-42]. It has been reported that EDTA enhances the availability of heavy metal to plants and thus increases the accumulation in their shoots [43-45].
Final Remarks and future research outlook
Basic and fundamental advances have been made in those last decades in understanding the processes implicated in metal phytoextraction from the contaminated soils. In the case of the chemically-assisted phytoextraction, the metal chelates dynamics in the rhizosphere have to be examined, either to overcome the risks associated with the low synthetic chelators degradability, or to optimize the use of more biodegradable compounds. In addition, researchers and policy makers have also to look for into the chemical pools of metals in soils and identify which ones are the targets for phytoextraction. A complete understanding of plant metal tolerance will be crucial to develop strategies to ameliorate the plant metal accumulation capacity. This will have significant involvements for phytoremediation. Since most of the known hyperaccumulator species are slow-growing and have small biomass, expressing their metal-accumulating genes in fast-growing, high biomass plants, is a promising approach for developing plants that can be used as novel tools in phytoextraction.
This work was in part financially supported by a grant from the Tunisian Ministry of Higher Education and Scientific Research contract program_2015-2018 CBS-LBAP/code: LR15CBS03.
- Kumar L, Talan A, Tyagi R (2020) Economic Development and Sustainability. Sustainability: Fundamentals and Applications 157-181. Link: https://bit.ly/3oIgtbO
- Székács A (2017) Environmental and ecological aspects in the overall assessment of bioeconomy. Journal of Agricultural and Environmental Ethics 30: 153-170. Link: https://bit.ly/2KbAQPU
- Anjum NA, Gill SS, Ahmad I, Pacheco M, Duarte AC, et al. (2012) The plant family Brassicaceae: An introduction, The plant family Brassicaceae. Springer 1-33. Link: https://bit.ly/376Mww2
- Palansooriya KN, Shaheen SM, Chen SS, Tsang DC, Hashimoto Y, et al. (2020) Soil amendments for immobilization of potentially toxic elements in contaminated soils: a critical review. Environ Int 134: 105046. Link: https://bit.ly/377AYZB
- Lewińska K, Karczewska A (2019) A release of toxic elements from military shooting range soils as affected by pH and treatment with compost. Geoderma 346: 1-10. Link: https://bit.ly/3oLVAN7
- Nascimento CWA D, Xing B (2006) Phytoextraction: a review on enhanced metal availability and plant accumulation. Scientia agricola 63: 299-311. Link: https://bit.ly/2Lswp3N
- Saad RB, Hsouna AB, Saibi W, Hamed KB, Brini F, et al. (2018) A stress-associated protein, LmSAP, from the halophyte Lobularia maritima provides tolerance to heavy metals in tobacco through increased ROS scavenging and metal detoxification processes. J Plant Physiol 231: 234-243. Link: https://bit.ly/2W0O4By
- Saibi W, Feki K, Yacoubi I, Brini F (2015) Bridging between proline structure, functions, metabolism, and involvement in organism physiology. Appl Biochem Biotechnol 176: 2107-2119. Link: https://bit.ly/37SaB9b
- Saibi W, Zouari N, Masmoudi K, Brini F (2016) Role of the durum wheat dehydrin in the function of proteases conferring salinity tolerance in Arabidopsis thaliana transgenic lines. Int J Biol Macromol 85: 311-316. Link: https://bit.ly/39X3ijj
- Sobhanian H, Pahlavan S, Meyfour A (2020) How does proteomics target plant environmental stresses in a semi-arid area? Mol Biol Rep 47: 3181-3194. Link: https://bit.ly/3qL6DI7
- Saibi W, Feki K, Mahmoud RB, Brini F (2015) Durum wheat dehydrin (DHN-5) confers salinity tolerance to transgenic Arabidopsis plants through the regulation of proline metabolism and ROS scavenging system. Planta 242: 1187-1194. Link: https://bit.ly/3qLVgQd
- Szczygłowska M, Piekarska A, Konieczka P, Namieśnik J (2011) Use of brassica plants in the phytoremediation and biofumigation processes. International Journal of Molecular Sciences 12: 7760-7771. Link: https://bit.ly/3ozlIL0
- Babula P, Adam V, Havel L, Kizek R (2012) Cadmium accumulation by plants of Brassicaceae family and its connection with their primary and secondary metabolism, The plant family Brassicaceae. Springer 71-97. Link: https://bit.ly/2IJ3bwA
- Hasanuzzaman M, Hossain MA, da Silva JAT, Fujita M (2012) Plant response and tolerance to abiotic oxidative stress: antioxidant defense is a key factor, Crop stress and its management: Perspectives and strategies. Springer 261-315. Link: https://bit.ly/2K8EOZr
- Dutta A, Patra A, Jatav HS, Jatav SS, Singh SK, et al. (2020) Toxicity of Cadmium in Soil-Plant-Human Continuum and Its Bioremediation Techniques, Soil Contamination. IntechOpen. Link: https://bit.ly/2W4smNe
- Ji P, Sun T, Song Y, Ackland ML, Liu Y (2011) Strategies for enhancing the phytoremediation of cadmium-contaminated agricultural soils by Solanum nigrum L. Environmental pollution 159: 762-768. Link: https://bit.ly/2JK6c09
- Sharma P, Tripathi S, Chandra R (2020) Highly efficient phytoremediation potential of metal and metalloids from the pulp paper industry waste employing Eclipta alba (L) and Alternanthera philoxeroide (L): Biosorption and pollution reduction. Bioresour Technol 319: 124-147. Link: https://bit.ly/2W3VRP4
- Chaney R (1983) Plant uptake of inorganic waste. Land treatment of hazardous wastes. Link: https://bit.ly/375ng9t
- Ali H, Khan E, Sajad MA (2013) Phytoremediation of heavy metals—concepts and applications. Chemosphere 91: 869-881. Link: https://bit.ly/37WE5CX
- Mahmood Q, Rashid A, Ahmad SS, Azim MR, Bilal M (2012) Current status of toxic metals addition to environment and its consequences, The Plant Family Brassicaceae. Springer 35-69. Link: https://bit.ly/2Kc2egv
- Gill SS, Anjum NA, Ahmad I, Thangavel P, Sridevi G, et al. (2012) Metal hyperaccumulation and tolerance in Alyssum, Arabidopsis and Thlaspi: an overview, The Plant Family Brassicaceae. Springer 99-137. Link: https://bit.ly/2W3Bh1h
- Bulak P, Walkiewicz A, Brzezińska M (2014) Plant growth regulators-assisted phytoextraction. Biologia plantarum 58: 1-8. Link: https://bit.ly/2W5U5gu
- Bareen F (2012) Chelate assisted phytoextraction using oilseed brassicas. Environ Pollut 21: 289-311. Link: https://bit.ly/377kIba
- Chaturvedi R, Favas P, Pratas J, Varun M, Paul MS (2019) EDTA-Assisted Metal Uptake in Raphanus sativus L. and Brassica oleracea L.: Assessment of Toxicity and Food Safety. Bull Environ Contam Toxico 103: 490-495. Link: https://bit.ly/3nlzpNz
- Sytar O, Ghosh S, Malinska H, Zivcak M, Brestic M (2020) Physiological and molecular mechanisms of metal accumulation in hyperaccumulator plants. Physiologia Plantarum. Link: https://bit.ly/344x5Tr
- Elallem KA, Sobeh M, Boularbah A, Yasri A (2020) Chemically degraded soil rehabilitation process using medicinal and aromatic plants. Environ Sci Pollut Res Int 1-21. Link: https://bit.ly/3oHSvNQ
- Kumar A, Subrahmanyam G, Mondal R, Cabral-Pinto M, Shabnam AA, et al. (2020) Bio-remediation approaches for alleviation of cadmium contamination in natural resources. Chemosphere 128855. Link: https://bit.ly/344xa9H
- Kumar A, Voropaeva O, Maleva M, Panikovskaya K, Borisova G, et al. (2020) Bioaugmentation with copper tolerant endophyte Pseudomonas lurida strain EOO26 for improved plant growth and copper phytoremediation by Helianthus annuus. Chemosphere 128983. Link: https://bit.ly/2W3E5eJ
- Ali Z, Waheed H, Gul A, Afzal F, Anwaar K, et al. (2017) Brassicaceae plants: Metal accumulation and their role in phytoremediation. Oilseed Crops: Yield and Adaptations under Environmental Stress 207-223. Link: https://bit.ly/3a1HGlC
- Saad RB, Farhat-Khemekhem A, Halima NB, Hamed KB, Brini F, et al. (2018) The LmSAP gene isolated from the halotolerant Lobularia maritima improves salt and ionic tolerance in transgenic tobacco lines. Funct Plant Biol 45: 378-391. Link: https://bit.ly/2W1qtAP
- Saibi W, Drira M, Yacoubi I, Feki K, Brini F (2015) Empiric, structural and in silico findings give birth to plausible explanations for the multifunctionality of the wheat dehydrin (DHN-5). Acta Physiologiae Plantarum 37: 52. Link: https://bit.ly/37UIdmY
- Ozyigit II, Can H, Dogan I (2020) Phytoremediation using genetically engineered plants to remove metals: a review. Environmental Chemistry Letters 1-30. Link: https://bit.ly/344xm8V
- Dixit A, Tomar P, Vaine E, Abdullah H, Hazen S, et al. (2018) A stress‐associated protein, AtSAP13, from Arabidopsis thaliana provides tolerance to multiple abiotic stresses. Plant Cell Environ 41: 1171-1185. Link: https://bit.ly/3oH57Vt
- Dixit AR, Dhankher OP (2011) A novel stress-associated protein ‘AtSAP10’from Arabidopsis thaliana confers tolerance to nickel, manganese, zinc, and high temperature stress. PLoS One 6: e20921. Link: https://bit.ly/2KbCMYG
- Jawad Hassan M, Ali Raza M, Ur Rehman S, Ansar M, Gitari H, et al. (2020) Effect of Cadmium Toxicity on Growth, Oxidative Damage, Antioxidant Defense System and Cadmium Accumulation in Two Sorghum Cultivars. Plants 9: 1575. Link: https://bit.ly/2JXcNEi
- Rizvi A, Ahmed B, Zaidi A, Khan MS (2020) Biosorption of heavy metals by dry biomass of metal tolerant bacterial biosorbents: an efficient metal clean-up strategy. Environ Monit Assess 192: 801. Link: https://bit.ly/37QtgCp
- Bidar G, Pelfrêne A, Louvel B, Janus A, Douay F (2019) Influence of amendments on metal environmental and toxicological availability in highly contaminated brownfield and agricultural soils. Environ Sci Pollut Res Int 26: 33086-33108. Link: https://bit.ly/3gzMEY4
- Zanin L, Tomasi N, Cesco S, Varanini Z, Pinton R (2019) Humic substances contribute to plant iron nutrition acting as chelators and biostimulants. Front Plant Sci 10: 675. Link: https://bit.ly/3mccnqO
- Nascimento CWA D (2006) Organic acids effects on desorption of heavy metals from a contaminated soil. Scientia Agricola 63: 276-280. Link: https://bit.ly/2Ln3QVf
- Bacaha N, Shamas R, Bakht J, Rafi A, Farhatullah GA (2015) Effect of heavy metal and EDTA application on plant growth and phyto-extraction potential of Sorghum (Sorghum bicolor). Pakistan Journal of Botany 47: 1679-1684. Link: https://bit.ly/3gAAs9D
- Kamran MA, Bibi S, Xu Rk, Hussain S, Mehmood K, et al. (2017) Phyto-extraction of chromium and influence of plant growth promoting bacteria to enhance plant growth. Journal of Geochemical Exploration 182: 269-274. Link: https://bit.ly/3gARqEQ
- Roshanfar M, Khanlarian M, Rashchi F, Motesharezadeh B (2020) Phyto-extraction of zinc, lead, nickel, and cadmium from a zinc leach residue. Journal of Cleaner Production 266: 121539. Link: https://bit.ly/37554Nq
- Grčman H, Velikonja-Bolta Š, Vodnik D, Kos B, Leštan D (2001) EDTA enhanced heavy metal phytoextraction: metal accumulation, leaching and toxicity. Plant and Soil 235: 105-114. Link: https://bit.ly/377rqhl
- He Q, Li H, Xu C, Sun Q, Bertness MD, et al. (2020) Consumer regulation of the carbon cycle in coastal wetland ecosystems. Philosophical Transactions of the Royal Society B 375: 20190451 . Link: https://bit.ly/344Y1SK
- Zanin L, Tomasi N, Cesco S, Varanini Z, Pinton R (2019) Humic substances contribute to plant iron nutrition acting as chelators and biostimulants. Front Plant Sci 10: 675. Link: https://bit.ly/3mccnqO
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.


 Save to Mendeley
Save to Mendeley
