Open Journal of Environmental Biology
Effect of Eager Grow-Up, A Glyphosate Based Herbicides, on Condition Factor and the Liver of Clarias Gariepinus
1Department of Biology and Forensic Science, Admiralty University, Ibusa, Delta State, Nigeria
2Niger-Delta University, Amassoma, Bayelsa State, Nigeria
3Distev Nigeria Limited, Nigeria
Author and article information
Cite this as
Bardi JI, et al. Effect of Eager Grow-Up, A Glyphosate Based Herbicides, on Condition Factor and the Liver of Clarias Gariepinus. Open J Environ Biol. 2026; 11(1): 8-14. Available from: 10.17352/ojeb.000053
Copyright License
© 2026 Bardi JI, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.This study investigated the effects of sub-lethal exposure to a glyphosate-based herbicide on water quality, hepatosomatic index (HSI), liver enzyme activity, and liver histopathology of Clarias gariepinus under laboratory conditions. Physico-chemical parameters of aquaria water showed only slight, non-significant variations between treatment and control groups and remained within acceptable limits for warm-water fish, indicating that observed effects were not attributable to water quality changes. A significant, time-dependent reduction in HSI was recorded in exposed fish, suggesting progressive hepatic stress. Serum activities of aspartate aminotransferase, alanine aminotransferase, and alkaline phosphatase increased with exposure duration, indicating impaired liver function. Histopathological examination revealed inflammatory cell infiltration, hepatocellular necrosis, hepatocyte depletion, and fatty degeneration, increasing over time. The combined physiological, biochemical, and histological responses demonstrate that the liver is a primary target organ of herbicide toxicity. These findings highlight the usefulness of integrating organosomatic indices, liver enzyme biomarkers, and histopathology in aquatic toxicity assessments and emphasize the potential ecological risks associated with herbicide contamination of aquatic environments.
Herbicides constitute a major group of agrochemicals widely employed to enhance agricultural productivity through effective weed control. Among these, glyphosate-based herbicides have gained extensive global acceptance due to their broad-spectrum efficacy and relative cost-efficiency [1,2]. The widespread application of glyphosate has, however, generated increasing concern regarding its environmental fate, persistence, and potential ecological risks, particularly in aquatic ecosystems [3,4]. Owing to its high water solubility and mobility, glyphosate readily enters surface waters through agricultural runoff, leaching, and soil erosion, thereby creating exposure pathways for non-target aquatic organisms and ecosystems [3,4].
In Nigeria, glyphosate-based herbicides are commonly used across diverse agro-ecological zones, often with limited regulatory oversight and inadequate awareness of associated environmental and health risks [5]. Such indiscriminate application increases the likelihood of contamination of surrounding water bodies. Recent environmental assessments have confirmed the occurrence of glyphosate residues in water, sediment, and fish samples from selected Nigerian rivers, indicating widespread contamination and potential ecological risks [6]. These findings align with earlier reports on the environmental fate of glyphosate in Nigerian aquatic ecosystems, highlighting its persistence and potential to adversely affect aquatic organisms [4]. Acute toxicity studies have further demonstrated that glyphosate exposure can negatively impact freshwater fish species, including African catfish juveniles, thereby posing risks to both fisheries sustainability and human consumers [7].
Fish are widely recognized as reliable bioindicators of aquatic ecosystem health due to their sensitivity to environmental stressors and their ecological and economic importance [8]. The African catfish (Clarias gariepinus) is among the most extensively cultured and consumed fish species in Nigeria, valued for its hardiness, rapid growth rate, and adaptability to varying environmental conditions. Because of these attributes, C. gariepinus has been extensively used in ecotoxicological studies to evaluate the biological effects of waterborne pollutants [4,9].
Previous studies have demonstrated that exposure to glyphosate-based herbicides induces significant histopathological alterations in vital fish organs. In C. gariepinus, sub-lethal glyphosate exposure has been associated with liver damage characterized by hepatocyte degeneration, necrosis, vacuolization, and vascular congestion [10,11]. Similar pathological responses have been linked to oxidative stress and disruption of antioxidant defense mechanisms in fish exposed to environmental contaminants [12,13]. Histopathological examination, therefore, serves as a sensitive and reliable tool for detecting early tissue-level responses to toxicant exposure, even at concentrations that do not cause immediate mortality [14,15].
The liver is a central organ responsible for metabolism, detoxification, and maintenance of physiological homeostasis in fish, rendering it particularly susceptible to chemical stressors [14,16]. Alterations in liver structure and function can compromise vital metabolic processes and overall fish health. Biomarkers such as hepatosomatic index (HSI) have been widely used to assess liver condition and metabolic stress in fish exposed to pesticides and other environmental contaminants [14,17]. Changes in HSI often reflect hepatic enlargement or degeneration resulting from increased detoxification demand or cellular damage [17].
In addition to internal organ pathology, morphometric indices such as condition factor are commonly used to assess the general health, nutritional status, and physiological fitness of fish populations. Condition factor, which relates body weight to length, provides insight into the well-being of fish and their response to environmental conditions [18]. Variations in condition factor have been associated with pollutant exposure, habitat degradation, and compromised water quality [19,20]. Studies conducted in Nigerian aquatic environments have shown that changes in condition factor of C. gariepinus are linked to environmental stress and water quality deterioration [20,21]. When combined with histopathological assessments, condition factor measurements offer a more comprehensive evaluation of contaminant-induced effects, linking organism-level responses to tissue-level damage.
Water quality parameters further play a critical role in mediating the effects of pollutants on fish health. Alterations in physicochemical characteristics such as dissolved oxygen, pH, and nutrient load can exacerbate the toxicity of agrochemicals in aquatic systems [22,23]. Poor water quality, coupled with chemical contamination, can intensify physiological stress and increase the susceptibility of fish to toxic insults [23].
Despite increasing evidence of glyphosate contamination in Nigerian aquatic environments and its documented toxicological effects on fish, there remains a paucity of studies that simultaneously evaluate both external health indices, such as condition factor, and internal liver pathology in Clarias gariepinus, particularly following exposure to specific glyphosate formulations such as Eager Grow-Up. Furthermore, limited information is available on whether the observed effects are dependent on exposure duration.
Therefore, this study investigates the impact of Eager Grow-Up, a glyphosate-based herbicide, on the condition factor and liver histopathology of the African catfish (Clarias gariepinus). The specific objectives are to determine the effect of the herbicide on the condition factor of the fish, to assess liver histopathological alterations induced by exposure, and to evaluate whether these effects are time-dependent.
Materials and methods
Experimental fish
A total of forty (40) apparently healthy African catfish (Clarias gariepinus) were used for this study. The fish had a mean body weight of 32.10 ± 0.20 g and a mean total length of 15.17 ± 0.15 cm. The specimens were obtained from a private fish farm located in Tombia Village, Bayelsa State, Nigeria. The fish were carefully transported in a Fifty litres trough to the laboratory in a well-aerated Fifty litres trough containing pond water to minimize stress and mortality.
Experimental site
The experiment was conducted in the Department of Biological Sciences Laboratory, Niger Delta University, Wilberforce Island, Bayelsa State, Nigeria.
Acclimatization of fish
Upon arrival at the laboratory, the fish were acclimatized individually in circular plastic aquaria for a period of five (5) days before the commencement of the experiment. During the acclimatization period, the aquaria were covered with mesh to prevent fish escape and external contamination. The fish were fed once daily with a commercial diet containing 35% crude protein, at a feeding rate of 1% of total biomass per day. Uneaten feed and waste materials were removed regularly to maintain water quality. No mortality was recorded during the acclimatization period.
Ethical considerations
All experimental procedures involving Clarias gariepinus were conducted in accordance with the ethical guidelines for the care and use of animals in research as approved by the Institutional Animal Ethics framework of Niger Delta University, Bayelsa State, Nigeria. The study was carried out as part of routine academic research, for which formal ethical approval numbers are not required, and all efforts were made to minimize the stress and suffering of the experimental animals.
Herbicide used
The toxicant used in this study was Eager Grow-Up, a glyphosate-based herbicide formulation. The herbicide stock solution had an original concentration of 41 g%, from which various test concentrations were prepared using distilled water.
Range-finding test
A preliminary range-finding test was conducted to determine suitable sub-lethal concentrations of the herbicide for the definitive test. Five concentrations were prepared by adding 0.04, 0.08, 0.15, 0.23, and 0.31 ml of the original herbicide solution to 30 litres of water in separate aquaria. Fish were exposed to these concentrations to observe behavioral responses and survival. The purpose of the range-finding test was to identify a concentration that would not cause immediate mortality but could elicit sub-lethal physiological and histological effects.
Experimental design (Definitive Test)
Based on the results of the range-finding test, a sub-lethal concentration of 0.20 mg/L (0.20 ppm) was selected for the definitive exposure experiment. The experimental design consisted of four treatment groups, each corresponding to a different exposure duration (24, 48, 72, and 96 hours), with each treatment having its own control group.
For each treatment and control group, three (3) fish were introduced individually into separate aquaria. Fish in the treatment groups were exposed to 0.20 ppm of Eager Grow-Up, while fish in the control groups were maintained in herbicide-free water under identical conditions. The toxicant was introduced into the aquaria using a graduated pipette to ensure accurate dosing.
Exposure duration and sample collection
At the end of each exposure period (24, 48, 72, and 96 hours), fish from the corresponding treatment and control groups were removed from the aquaria. The fish were humanely sacrificed, and the liver tissues were carefully excised for histological analysis. Each liver sample was appropriately labeled according to treatment group and exposure duration.
Determination of liver enzymes (ALT, AST, ALP)
Blood samples were collected from the caudal vein of each fish and allowed to clot at room temperature. Serum was separated by centrifugation at 3000 rpm for 10 minutes. Serum Alkaline Phosphatase (ALP) activity was determined following the method of [24], while Serum Aspartate Aminotransferase (AST) and Alanine Aminotransferase (ALT) activities were measured according to the method of [25]. Enzyme activities were read spectrophotometrically at the specified wavelengths and expressed in units per liter (U/L).
Physicochemical parameters of water
Physicochemical properties of the aquarium water were monitored throughout the experimental period. Parameters assessed included pH, Conductivity, Temperature, Turbidity, Dissolved oxygen, and Alkalinity. The physicochemical properties such as turbidity, temperature, conductivity, and pH were analysed using an in-situ meter, dissolved oxygen was analysed using the wrinklers method, and alkalinity was analysed by the titration method.
Sample collection of the liver
After each time duration, the liver of the fish was collected after sacrificing the fish and placed in well-labeled formalin sample bottles for histopathological examination.
Histological analysis of liver
The excised liver tissues were processed for histological examination using standard histological techniques. The tissues were fixed, dehydrated, cleared, embedded in paraffin wax, sectioned using a microtome, and stained for microscopic examination. Histopathological alterations in liver architecture were observed and documented to assess the hepatological effects of the glyphosate-based herbicide on Clarias gariepinus.
Statistical analysis
Results were expressed as mean ± standard deviation (SD). Differences between control and treatment groups at the various exposure durations (24, 48, 72, and 96 hours) were analyzed using one-way analysis of variance (ANOVA). When significant differences were detected, pairwise comparisons with Bonferroni correction were performed to identify specific differences among group means. Significant differences were indicated using superscript letters (a, b, ab). A probability level of p < 0.05 was considered statistically significant. All analyses were performed using PAST software (Paleontological Statistics), version 4.04.
Results
The physicochemical parameters of the aquarium water exposed to a sub-lethal concentration of the herbicide at varying exposure durations are presented in Table 1. The mean values of most water quality parameters showed slight but statistically significant variations (p < 0.05) between the treatment and control groups. However, conductivity and turbidity remained unchanged, exhibiting similar values in both the exposed and control aquaria throughout the experimental period.
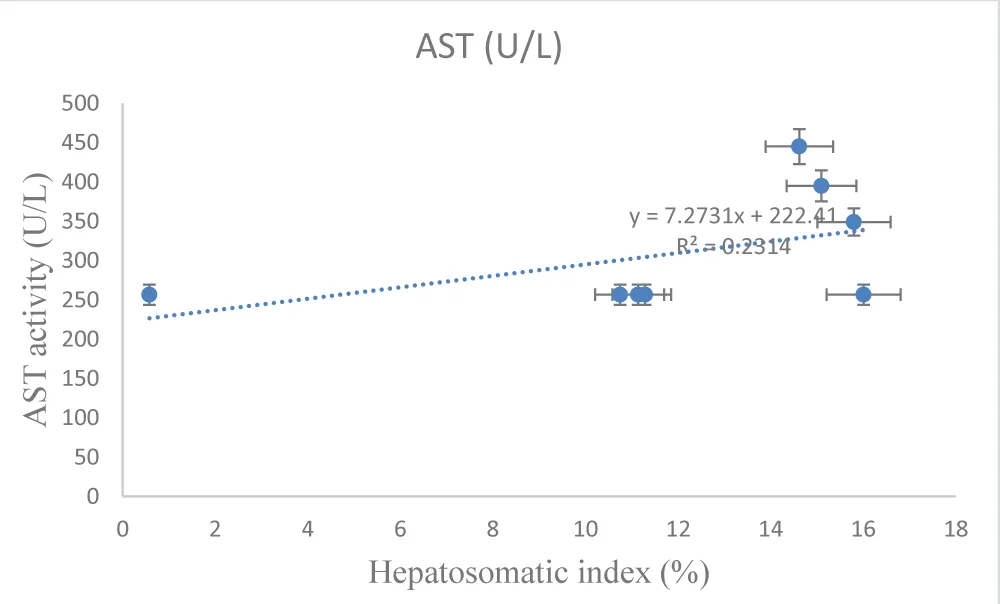
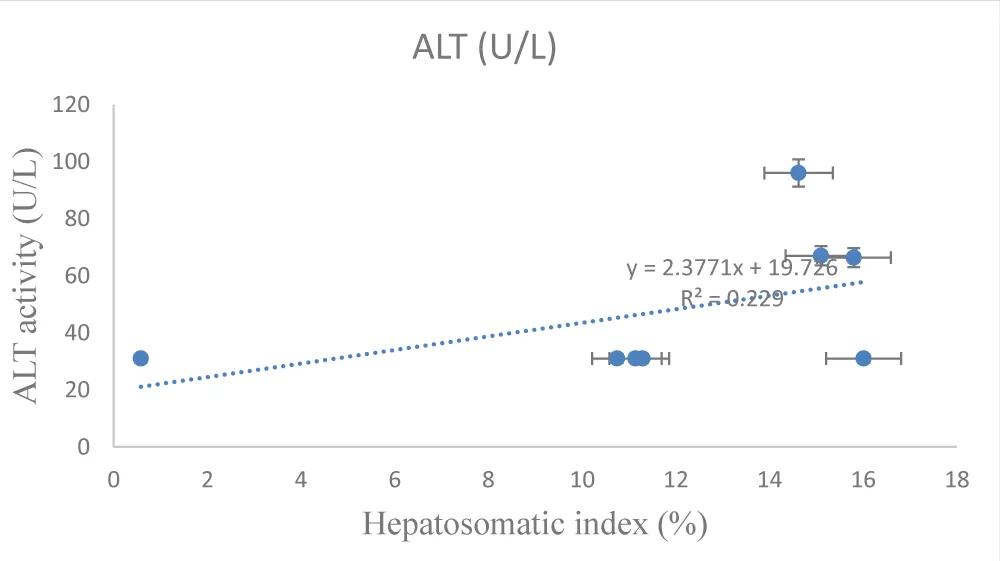
Exposure of Clarias gariepinus to sub-lethal concentrations of Eager Grow-Up (glyphosate) caused time-dependent changes in liver physiology and biochemistry. The Hepatosomatic Index (HSI) was consistently higher in treated fish compared to controls, indicating liver enlargement. Liver enzymes—AST, ALT, and ALP-increased progressively over time in the treatment group, with the highest values observed at 96 hours, suggesting hepatic stress and possible liver damage. The condition factor remained relatively stable, indicating that overall body condition was minimally affected during the exposure period (Tables 2,3) [Figures 1-3].
Discussion
Water quality is a critical determinant of experimental reliability in aquatic toxicology, as fluctuations in physico-chemical parameters can independently influence fish physiology and confound toxicant-induced effects. In the present study, key water quality parameters of aquaria exposed to sub-lethal concentrations of the herbicide were monitored alongside control systems. Although slight fluctuations were observed between treatment and control groups, these variations were minimal and statistically insignificant, with all measured parameters remaining within established tolerance limits for warm-water fish species. Comparable acceptable ranges have been reported by [11,22,23], and more recently by [26], who emphasized that maintaining stable water quality minimizes secondary stress responses in toxicological experiments. Consequently, the observed biological, biochemical, and histopathological alterations in the exposed fish can be attributed primarily to herbicide exposure rather than to changes in water quality.
The liver plays a central role in xenobiotic metabolism and detoxification in fish, functioning as the primary site for biotransformation of chemical contaminants into less toxic and more readily excretable metabolites. Owing to this role, hepatic tissues are widely used as sensitive biomarkers for assessing pollutant-induced stress in aquatic organisms [13,16]. The hepatosomatic index (HSI), which represents the ratio of liver weight to total body weight, provides an integrated measure of hepatic response to environmental stressors and toxicants [12,19].
The results presented in Table 2 demonstrate that exposure to sub-lethal concentrations of Eager Grow-Up (glyphosate) induced time-dependent changes in liver physiology and biochemical parameters. The HSI of treated fish was consistently higher than that of controls at early exposure (24 h) but gradually declined over time, suggesting progressive hepatic stress. In parallel, liver enzyme activities (AST, ALT, and ALP) increased steadily with exposure duration, with the highest values recorded at 96 hours. These elevations indicate hepatocellular stress and potential liver damage, as these enzymes are well-established biomarkers of liver function [24,25]. The progressive rise in enzyme levels reflects the cumulative toxic effect of glyphosate on hepatic metabolism [27], while the relatively stable condition factor (CF) suggests that overall somatic growth was minimally affected during the exposure period [28-31]. Together, these physiological and biochemical responses align with the histopathological observations, highlighting the sensitivity of liver biomarkers in detecting sub-lethal herbicide-induced toxicity.
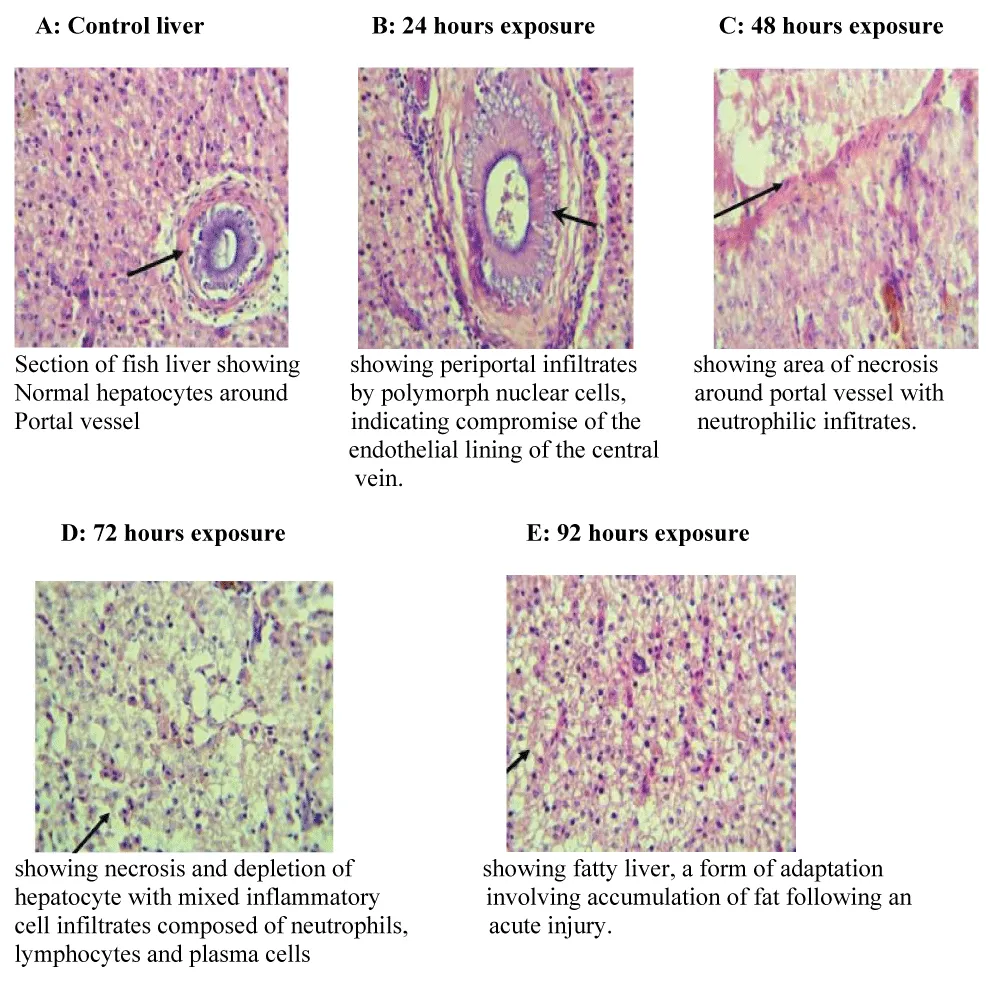
Histopathological examination further corroborated the adverse effects of the herbicide on hepatic integrity. Liver sections from control fish displayed normal architecture, with well-organized hepatocytes surrounding the portal vessels. In contrast, exposed fish exhibited progressive pathological alterations with increasing exposure duration. After 24 hours, periportal infiltration by polymorphonuclear cells was evident, suggesting early inflammatory responses and endothelial compromise. At 48 hours, focal necrosis around portal areas accompanied by neutrophilic infiltration was observed, indicating hepatocellular damage. The 72-hour exposure group showed extensive necrosis, hepatocyte depletion, and mixed inflammatory cell infiltration, reflecting advanced tissue injury. By 96 hours, the liver architecture was characterized by sheets of hepatocytes with clear cytoplasm and thin fibrous stroma, consistent with hepatic steatosis (fatty liver), a known adaptive response to acute toxic insult involving lipid accumulation due to impaired lipid metabolism.
These findings are consistent with earlier reports by [15], who observed increased lipid droplet size and hepatic vacuolization in fish exposed to atrazine, and by [11], who reported leukocyte infiltration, hepatocyte hypertrophy, pyknotic nuclei, and cytoplasmic vacuolization in fish exposed to glyphosate. More recent studies have similarly demonstrated that herbicides and other agrochemicals induce oxidative stress, inflammatory responses, and lipid metabolic disorders in fish livers, leading to steatosis and necrosis [13,14,16].
Overall, the combined evidence from water quality assessment, hepatosomatic index analysis, liver enzyme activity (Table 2), and histopathological observations clearly indicates that sub-lethal exposure to the herbicide elicits time-dependent hepatic toxicity in Clarias gariepinus. The liver responses observed in this study underscore the suitability of hepatic biomarkers as early warning indicators of environmental contamination and highlight the ecological risks associated with herbicide contamination of aquatic ecosystems.
Recommendations
Regular monitoring and stricter regulation of herbicide application near aquatic environments are recommended to minimize contamination and protect non-target fish species. Hepatosomatic indices, liver enzyme analyses, and histopathology should be routinely employed as sensitive biomarkers for early detection of sub-lethal toxic effects in aquatic toxicological assessments. Further studies incorporating long-term exposure, recovery experiments, and molecular biomarkers are necessary to better understand the mechanisms and chronic impacts of herbicide toxicity. Public education on environmentally responsible agrochemical use should be strengthened to reduce herbicide runoff and safeguard aquatic ecosystems.
- Dill GM, CaJacob CA, Padgette SR. Glyphosate-resistant crops: Adoption, use, and future considerations. Pest Manag Sci. 2010;66(4):326–331. Available from: https://doi.org/10.1002/ps.1501
- Feng PCC, CaJacob CA, Martino-Catt SJ, Cerny RE, Elmore GA, Heck GR, et al. Glyphosate-resistant crops: Developing the next generation products. In: Nandula VK, editor. Glyphosate resistance in crops and weeds. Hoboken (NJ): John Wiley & Sons; 2010. p. 45–65. Available from: https://doi.org/10.1002/9780470634394.ch3
- Battaglin WA, Meyer MT, Kuivila KM, Dietze JE. Glyphosate and its degradation product AMPA in soils, surface water, groundwater, and precipitation in the USA. J Am Water Resour Assoc. 2014;50(2):275–290. Available from: https://doi.org/10.1111/jawr.12159
- Ujowundu CO, Onyishi GC, Eze JI. Environmental fate and ecological effects of glyphosate-based herbicides in Nigerian aquatic ecosystems. Afr J Environ Sci Technol. 2017;11(9):417–426. Available from: https://doi.org/10.5897/AJEST2017.2356
- Al Ghanim KA, Al Ghanim MK, Al Humaidan AA. Herbicide-induced hepatotoxicity and oxidative stress in freshwater fish. Environ Toxicol Pharmacol. 2019;69:154–162.
- Alarape SA, Fagbohun ED, Ipadeola AO, Adeigbo AA, Adesola OO, Adeyemo OK. Occurrence of glyphosate-based herbicide residues in water, sediment, and fish from selected rivers in Nigeria and associated ecological risks. Environ Monit Assess. 2023;195:1123. Available from: https://doi.org/10.1007/s10661-023-11789-5
- Yerima R, Jacob LT, Nazeef S. Acute toxicity of glyphosate and propanil on Clarias gariepinus juveniles. Greener J Biol Sci. 2023;13(1):16–23. Available from: https://doi.org/10.5281/zenodo.8141211
- Van der Oost R, Beyer J, Vermeulen NPE. Fish bioaccumulation and biomarkers in environmental risk assessment: A review. Environ Toxicol Pharmacol. 2003;13(2):57–149. Available from: https://doi.org/10.1016/S1382-6689(02)00126-6
- Edori O. Effects of sub-lethal exposure to monocrotophos on biochemical and histopathological indices in African catfish (Clarias gariepinus). J Aquat Sci. 2007;22(1):45–52.
- Akinbadewa A, Olurin KB, Ayoola SO. Histological alterations in organs of African catfish (Clarias gariepinus) exposed to sub-lethal concentrations of glyphosate. Borneo J Resour Sci Technol. 2021;11(2):3815. Available from: https://doi.org/10.33736/bjrst.3815.2021
- Ayoola SO. Histopathological effects of glyphosate on juvenile African catfish (Clarias gariepinus). Am-Eurasian J Agric Environ Sci. 2008;4(3):362–367. Available from: https://www.researchgate.net/publication/237539217
- Abdel-Daim MM, Abushouk AI, Aleya L. Hepatotoxicity biomarkers and oxidative stress responses in fish exposed to environmental contaminants. Environ Sci Pollut Res Int. 2020;27:45082–45096. Available from: https://doi.org/10.1007/s11356-020-11142-0
- Alimba CG, Faggio C. Biomarkers of oxidative stress and environmental pollutants in fish. Rev Aquac. 2019;11(3):1392–1416. Available from: https://doi.org/10.1111/raq.12284
- Banaee M, Mirvaghefi A, Ahmadi K. Biomarkers of liver function and hepatosomatic index in fish exposed to pesticides: A review. Environ Toxicol Pharmacol. 2019;68:1–13. Available from: https://doi.org/10.1016/j.etap.2019.05.001
- Biagianti-Risbourg S, Bastide J. Hepatic perturbation induced by atrazine in juvenile grey mullet Liza ramada: An ultrastructural study. Aquat Toxicol. 1995;31(3):217–229.
- Velki M, Lackmann C, Barranco A, Hollert H, Seiler TB. Whole-organism fluorescence as a biomarker for oxidative stress in Danio rerio larvae. Chemosphere. 2018;197:200–209. Available from: https://doi.org/10.1016/j.chemosphere.2018.01.045
- Abdel-Tawwab M, Monier MN, El-Sayed AFM. Hepatosomatic and organosomatic indices as indicators of liver stress in Nile tilapia (Oreochromis niloticus) under chemical exposure. Environ Sci Pollut Res Int. 2021;28:12345–12355. Available from: https://doi.org/10.1007/s11356-021-13456-7
- Froese R. Cube-law, condition factor and weight–length relationships: History, meta-analysis and recommendations. J Appl Ichthyol. 2006;22(4):241–253. Available from: https://doi.org/10.1111/j.1439-0426.2006.00805.x
- Inyang O, Ugwumba A, Eyo J. Condition factor, hepatosomatic and organosomatic indices of Clarias gariepinus exposed to sub-lethal concentrations of toxicants. Glob J Life Sci. 2017;13(2):11–18.
- Wakil UB, Mohammed GA, Ndirmbita WL, Bala HM. Biological water quality and condition factor of Clarias gariepinus in Lake Alau, Nigeria. Int J Nat Resour Ecol Manag. 2016;1(1):1–10.
- Adebambo AAR, Fafioye OO, Babarinde TD. Condition factor and observable disease manifestation of Clarias gariepinus obtained from non-wild ponds in Ibadan, south-western Nigeria. Jewel J Sci Res. 2025;10(1):93–101. Available from: https://journals.fukashere.edu.ng/index.php/jjsr/article/view/769
- Boyd CE. Water quality in warmwater fish ponds. Auburn (AL): Auburn University; 1979. Available from: https://www.scirp.org/reference/referencespapers?referenceid=2540718
- Seiyaboh EI, Ogamba EN, Utibe DI, Sikoki FD. Impact of dredging on water quality and fisheries of Igbedi Creek, Nigeria. IOSR J Environ Sci Toxicol Food Technol. 2013;7(5):38–44.
- Rec GSC. Colorimetric method for serum alkaline phosphatase determination. J Clin Chem Clin Biochem. 1972;10:182–184.
- Reitman S, Frankel S. Colorimetric method for determination of serum glutamic oxaloacetic and glutamic pyruvic transaminases. Am J Clin Pathol. 1957;28:56–63. Available from: https://doi.org/10.1093/ajcp/28.1.56
- Sathishkumar P, Krishnakumar R, Senthilkumaran B. Biochemical responses of fish liver to sub-lethal pesticide exposure. J Environ Biol. 2008;29(4):523–529.
- Ogunbiyi OA, Balogun JK, Olusanya OT. Hepatic biomarkers and condition indices in Clarias gariepinus following chronic agrochemical exposure. Afr J Aquat Sci. 2019;44(1):15–24.
- Abdel Tawwab M, El Sayed GO, Shady SH. Growth, biochemical variables, and zinc bioaccumulation in Nile tilapia (Oreochromis niloticus) as affected by waterborne zinc toxicity and exposure period. Int Aquat Res. 2016;8(3):197–206. Available from: https://link.springer.com/article/10.1007/s40071-016-0135-0
- Adeyemo FA, Okunowo OO, Arowolo TA. Assessment of glyphosate herbicide toxicity on somatic and biochemical indices in freshwater fish species. J Aquat Toxicol Environ. 2010;5(2):85–95.
- Ayanda IO, Adebayo EA, Eze SOO. Herbicide usage patterns and awareness of environmental and health risks among farmers in Nigeria. J Environ Sci Health B. 2021;56(6):486–495. Available from: https://doi.org/10.1080/03601234.2021.1896234
- Ezejiofor CR, Ufodike EBC, Nwankwo DE. Effects of glyphosate-based herbicide on liver biochemical parameters of African catfish. J Fish Aquat Sci. 2021;16(3):178–187. Available from: https://ijsred.com/volume4/issue3/IJSRED-V4I3P201.pdf
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.


 Save to Mendeley
Save to Mendeley
