Annals of Limnology and Oceanography
Possible anti-fouling effect of polyenes in molluscs shell
AV Zhirmunsky National Scientific Center of Marine Biology, Far Eastern Branch, Russian Academy of Science, Vladivostok, Russia
Author and article information
Cite this as
Karpenko M, Karpenko A, Odintsov V (2024) Possible anti-fouling effect of polyenes in molluscs shell. Ann Limnol Oceanogr. 2024; 9(1): 1-4. Available from: 10.17352/alo.000015
Copyright License
© 2024 Karpenko M, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Polyenes were analysed in two species of marine gastropods (the periwinkles Littorina brevicula and Littorina mandshurica) and two species of marine bivalves (the mussels Mytilus trossulus and Crenomytilus grayanus) to determine the relationship of these compounds with the fouling of molluscs shell. It was found that the shells of the Littorina gastropods and M. trossulus, which are rarely overgrown by fouling organisms in nature, contained polyenes. In rare cases of fouling by Balanus barnacles, there were no polyenes at the sites of their attachment on the shells. Polyenes were not found in the shells of C. grayanus, which are often prone to fouling. The role of polyenes in the possible anti-fouling properties of molluscan shells is discussed.
Integumentary colour is a fundamental aspect of the physiology and functional morphology of integuments that has probably played a crucial role in all stages of their evolution. Colour patterns serve for crypsis, an advertisement for mates, aposematism (warning), and startling prey, and also have non-communication functions like thermoregulation and resistance to mechanical wear. Colours are produced by a variety of pigment molecules, nanostructures, or a combination of both. This mechanistic diversity provides complex opportunities for natural and sexual selection on the many functions of the integumentary colour [1]. Polyenes play a substantial role as pigments in several molluscan taxa. Using Raman micro-spectrometry, these compounds have been identified as pigments in coloured parts of shells in 13 gastropods, one cephalopod, and four bivalve taxa. There are no trivial relationships between colour, pigment, and taxon. The same colour in different taxa may be due to different pigments; different colours in the same taxon are due to different pigments [2]. Based on an analysis of 89 polyene-based pigments in 45 molluscan species, Ishikawa, et al. [3] found that different pigments (from yellow, orange, and red to purple) are associated with polyenes characterised by different conjugation lengths (from 7–9, 9–10 to 10–12, respectively). Polyenes with less than seven conjugation lengths exhibit no visible colour because their absorption maxima are outside the visual light range [3]. The latter, as well as their antifungal properties, suggests that the role of polyenes may be reduced to providing not only shellfish colour, but also bioprotective functions. Fungicidin, the first known antifungal polyene subsequently referred to as nystatin, was isolated as a fermentation product of Streptomyces noursei cultured from a soil sample in 1949 [4]. In 1953, two active compounds—amphotericin A and amphotericin B (AmB), named for their amphoteric properties—were also isolated from Streptomyces nodosus. These polyenes were chemically similar to nystatin. Depending on the number of conjugated double bonds, polyenes are classified into trienes, tetraenes, pentaenes, hexaenes, heptaenes, etc. The tetraene amphotericin A showed an antifungal activity similar to that of nystatin, while the antifungal activity of heptaene AmB was significantly higher compared to nystatin and amphotericin A [5].
Taking into account the antifungal activity of polyenes, as well as the antifouling activity of bacterial extracts containing polyenes [5-7], we suggest that the fact that the clean, free-of-fouling surface of the shells in the molluscs under study may be explained by polyenes, which until recently have been investigated mainly as substances that determine the shell colour. The present study aimed to analyse several types of molluscan shells in order to confirm or refute this suggestion.
Materials and methods
Animals
To study the polyenes in molluscan shells we collected two species of marine gastropods (the periwinkles Littorina brevicula (Philippi 1844) and Littorina mandshurica (Schrenk, 1861)) and two species of marine bivalves (the mussels Mytilus trossulus (Gould, 1850) and Crenomytilus grayanus (Dunker, 1853)) in the Amur Bay, Peter the Great Bay, Sea of Japan. 7 Littorina mandshurica, 12 Littorina brevicula, 12 Crenomytilus grayanus and 18 Mytilus trossulus were examined.
Raman spectroscopy
The Raman spectra were recorded on an inVia Reflex Raman microspectrometer (Renishaw, United Kingdom) coupled with a Leica DM2500M incident light microscope (Leica Microsystems, Germany). For excitation, we used a 532-nm diode-pumped laser at a power of 1.0 mW and exposure time of 0.1 s and 100 – 200 scans. A laser spot with a diameter of ~1.4 μm on the sample was formed with a lens (50×, NA = 0.75, Leica). The spectra were processed using smooth, baseline subtraction, and peak fit functions in the WiRE 4.1 software. For peak fitting, a mixture of Gaussian and Lorentzian algorithms was used. The spectrum of each object at each location was measured over a range of 170 to 1800 cm–1.
Results and discussion
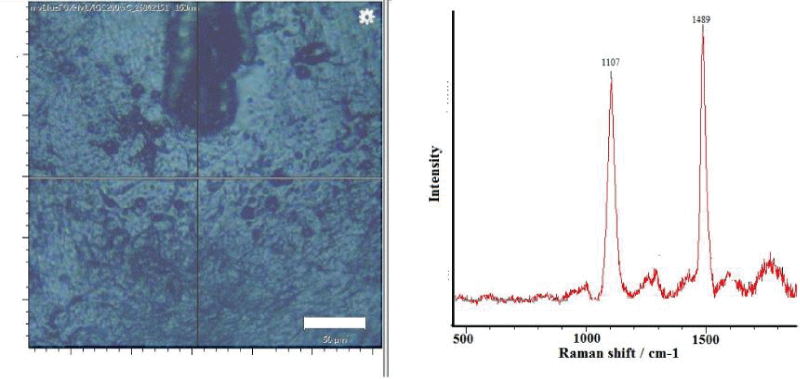
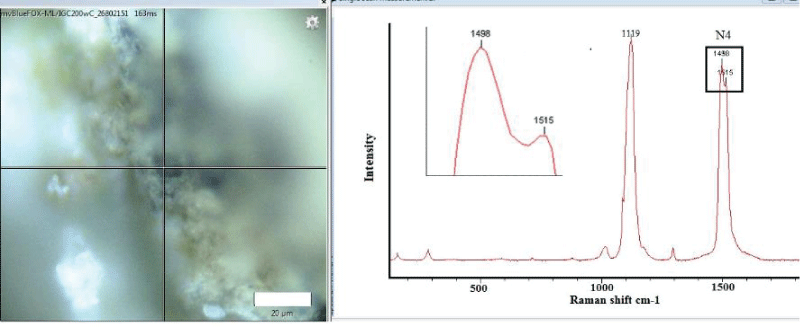
A Raman spectroscopy of the M. trossulus shells that did not have signs of fouling showed the presence of polyenes. Figure 1 shows a typical pattern where visual fouling (barnacles Balanus sp. etc.) is absent and Raman signals characteristic of polyenes are present. Two strong bands are observed: one corresponding to C = C stretching near 1489 cm-1 and another at 1107 cm-1. Number of double bonds, estimated using the formula (1) applied by Stemmer and Nehrke [8], is 16.
N4 = 830/(R4-1438) (1)
where: N4 - number of double bonds, R4 - raman shift of double bond
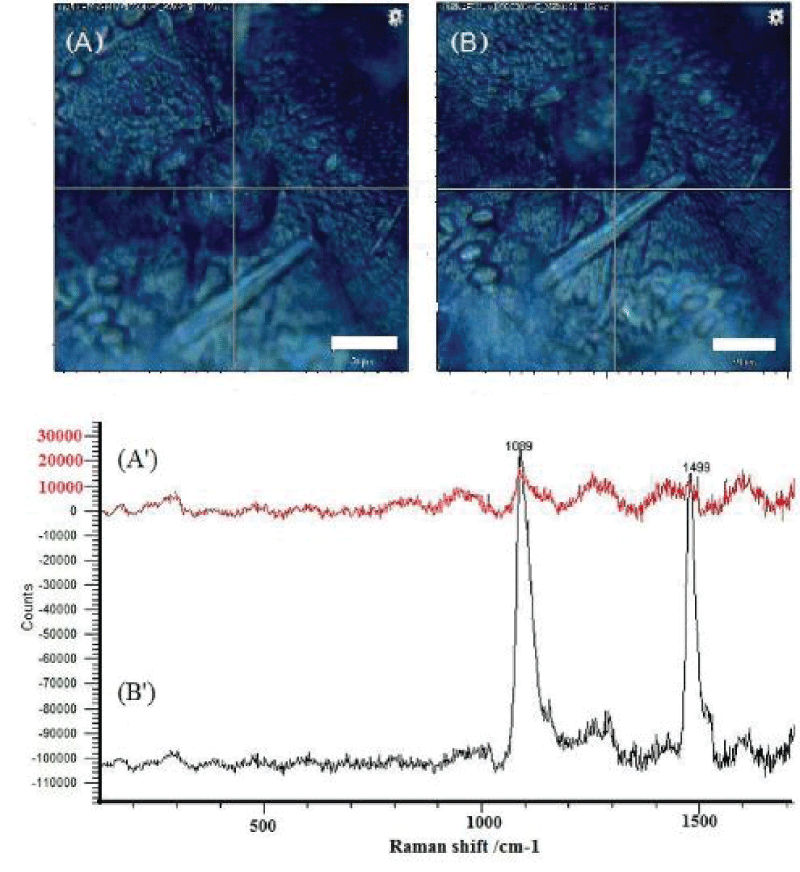
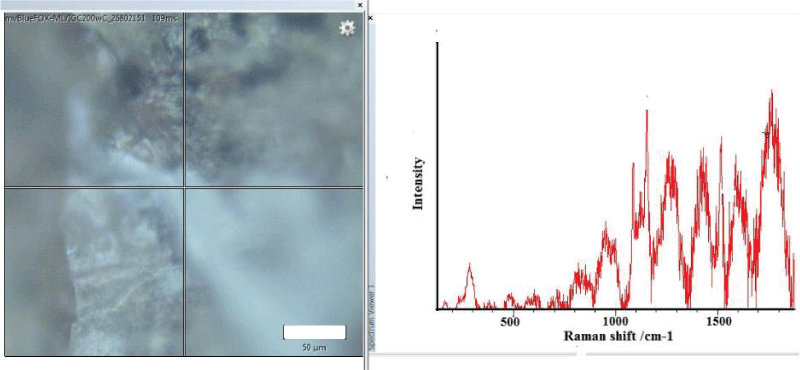
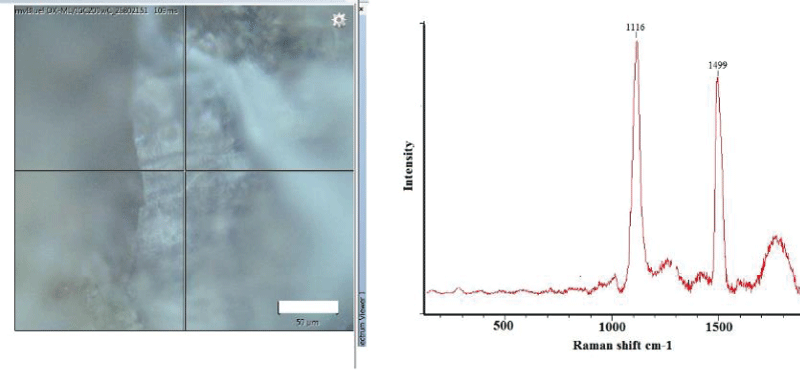
In the M. trossulus overgrown by barnacles, the same pattern was observed. However, a more detailed study showed the absence of polyenes in the shell at the point of attachment of barnacles that had settled in the cypris larva stage (Figure 2A). At a small distance (about 50 µm) from the attachment point, the signal from polyenes reappeared, representing polyenes with 14 double bonds (Figure 2B).
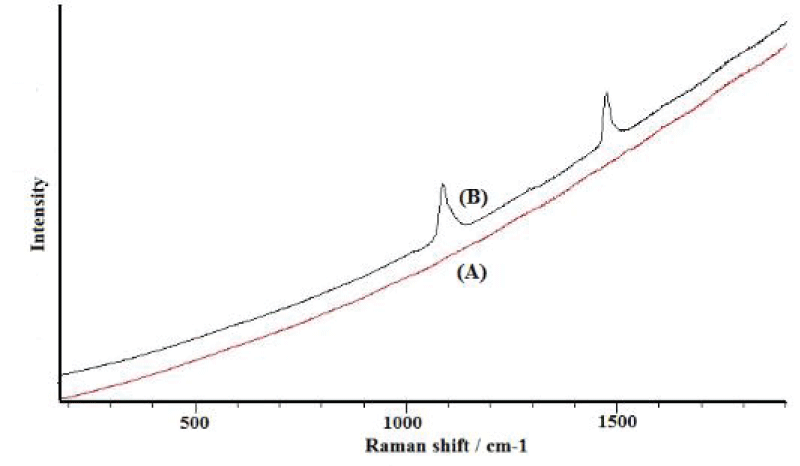
All the C. grayanus analysed were heavily overgrown by calcareous algae, bryozoans, and other epibionts. It was impossible to find any single specimen of C. grayanus free of fouling, and Raman spectroscopy of their shells showed the absence of polyenes in them (Figure 3A). The Raman spectrum for M. trossulus is shown (Figure 3B) for comparison.
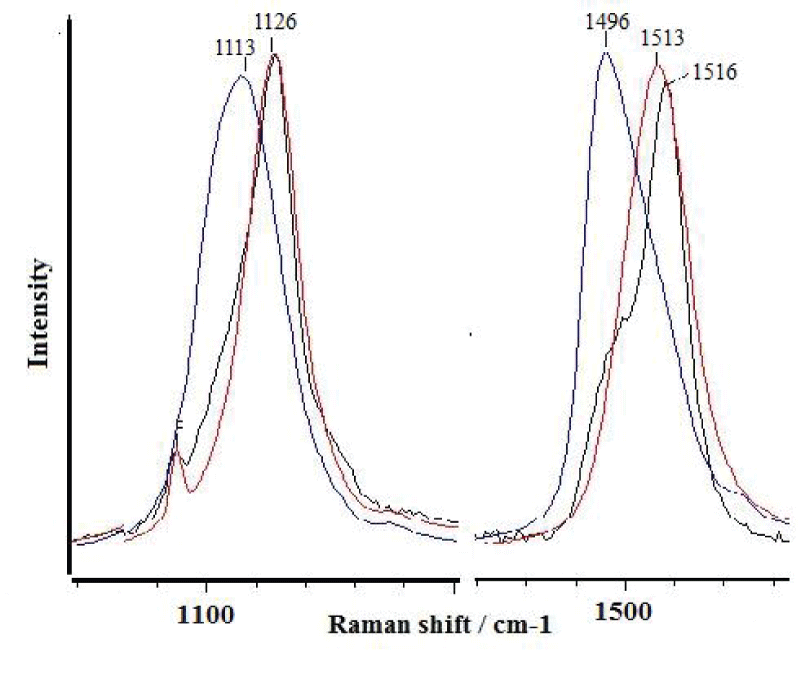
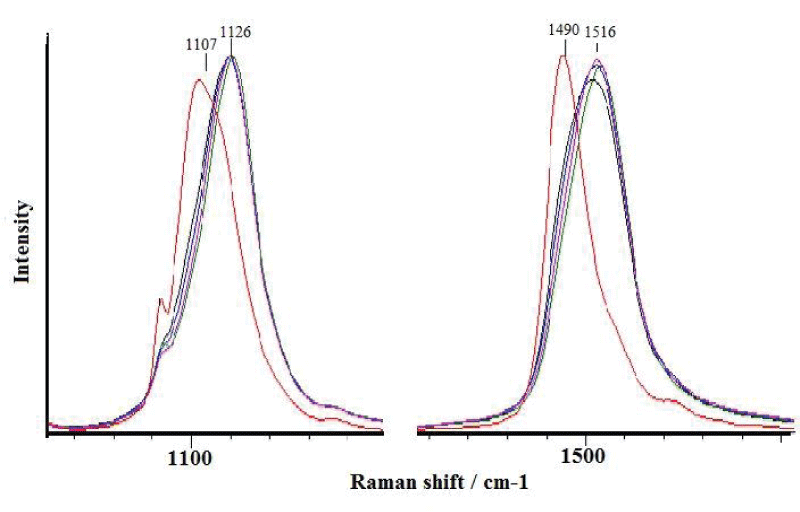
Shells of the studied Littorina gastropods are rarely affected by fouling, and all of the examined specimens showed the presence of polyenes. Among the collected L. brevicula, no specimens with attached barnacles were found. The Raman spectra at different points in L. brevicula shells show the presence of various polyenes (Figure 4). Polyenes are present regardless of the presence or lack of fouling. However, for a specimen overgrown by calcareous algae, the position of the characteristic peaks is shifted to a longer wavelength side (1107 and 1490 cm-1 vs. 1119 and 1510 cm-1, respectively) than for a non-fouled specimen, which indicates the presence of various polyenes with different numbers of conjugated double bonds, from 16 to 12, the same as in fouled and non-fouled shells (Figure 5).
A study of the shells of L. mandshurica has shown that some of the characteristic peaks are composite peaks that indicate the presence of several polyenes. Figure 6 presents a magnified part of the peak in the 1500 cm-1 region actually consisting of two peaks, 1498 and 1516 cm-1, which can indicate the presence of polyenes with estimated numbers of double bonds of 14 and 11, respectively.
As in the case of M. trossulus, polyenes were not found in the L. mandshurica shell surface at the barnacle’s attachment points. Figure 7 shows a Raman spectrum for the L. mandshurica shell surface at the point of the barnacle’s attachment. No essential modes associated with polyenes have been recorded. However, a point at 50 µm from the point of the barnacle’s attachment on the L. mandshurica shell surface contains polyenes, which is clearly visible in the Raman spectrum in Figure 8.
The authors do not claim that polyenes completely protect mollusk shells from fouling. More detailed research on a wider range of species is required as well as obtaining polyene extracts and directly determining their antifouling activity. Also, fouling classification is required to find any correlation with different polyenes distributions. However, the results indicate that antifouling may be one of the functions of polyenes, which until now have only attracted researchers as the cause of shell color. Considering the harm fouling causes to shellfish cultivation, reducing production yield [9-11], research in this area is of great importance, providing another parameter that should be taken into account when choosing shellfish for cultivation.
Conclusion
Observations of tropical molluscs have shown that their brightly coloured shells, in contrast to the molluscs from temperate waters considered in the present study, are usually free of fouling. The assumption that polyenes, besides pigmentation, have an antifouling function seems to be plausible, especially considering that most of the pigments in molluscan shells are polyenes of which some have antifungal properties and are colourless. However, our study does not fully confirm this. Only C. grayanus represents an example of heavily fouled shells with a complete lack of polyenes on the surface. Among the examined gastropods and bivalves, with the exception of C. grayanus, there were shells covered by fouling despite the presence of polyenes in general. Among the druse of the bivalve M. trossulus, there are both druse that (in the fewest cases) are highly susceptible to fouling and those (in most cases) with a clean shell. The fact that barnacle attachment sites on molluscs’ shells lack polyenes suggests that the latter generally counteracts fouling.
Author contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Maxim A. Karpenko, Alexander A. Karpenko, and Vyacheslav S. Odintsov. The first draft of the manuscript was written by Vyacheslav S. Odintsov and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript. All authors agree to be accountable for all aspects of the study in ensuring that questions related to the accuracy or integrity of any part of the study are appropriately investigated and resolved.
Funding
The authors declare that no funds, grants, or other special support were received during the preparation of this manuscript. This study was carried out at the A.V. Zhirmunsky National Scientific Center of Marine Biology, FEB RAS.
- Shawkey MD, D'Alba L. Interactions between colour-producing mechanisms and their effects on the integumentary colour palette. Philos Trans R Soc Lond B Biol Sci. 2017 Jul 5;372(1724):20160536. doi: 10.1098/rstb.2016.0536. PMID: 28533449; PMCID: PMC5444072.
- Hedegaard C, Bardeau JF, Chateigner D. Molluscan shell pigments: an in situ resonance Raman study. J. of Molluscan Studie. 2006; 72:157–162. https://doi.org/10.1093/mollus/eyi062
- Ishikawa M, Kagi H, Sasaki T, Endo K. Chemical basis of molluscan shell color revealed with in situ micro-Raman spectroscopy. J. of Raman Spectroscopy. 2019; 50:1700-1711. https://doi.org/10.1002/jrs.5708
- HAZEN EL, BROWN R. Fungicidin, an antibiotic produced by a soil actinomycete. Proc Soc Exp Biol Med. 1951 Jan;76(1):93-7. doi: 10.3181/00379727-76-18397. PMID: 14816400.
- Dutcher JD. The discovery and development of amphotericin B. Dis Chest. 1968 Oct; 54: Suppl 1:296-8. doi: 10.1378/chest.54.supplement_1.296. PMID: 4877964.
- Kharchenko U, Beleneva I, Izotov N, Vyalyi I, Egorkin V, Tsvetnikov A, Karpenko A, Nguyen V Chi. Preparation of PEO/polymer coating on aluminum alloy with antifouling properties. J. Coat. Technol. Res. 2022; 20:763–779. https://doi.org/10.1007/s11998-022-00706-x
- Chandrasekar P. Management of invasive fungal infections: a role for polyenes. J Antimicrob Chemother. 2011 Mar;66(3):457-65. doi: 10.1093/jac/dkq479. Epub 2010 Dec 14. PMID: 21172787.
- Stemmer K, Nehrke G. The distribution of polyenes in the shell of Arctica islandica from North Atlantic localities: a confocal Raman microscopy study. J. of Molluscan Studies. 2014; 80:365–370. https://doi.org/10.1093/mollus/eyu033
- Sievers M, Fitridge I, Dempster T, Keough MJ. Biofouling leads to reduced shell growth and flesh weight in the cultured mussel Mytilus galloprovincialis. Biofouling. 2013;29(1):97-107. doi: 10.1080/08927014.2012.749869. PMID: 23256892.
- Babarro JMF, Padin XA, Filgueira R, El Morabet H, Portabales MAL. The impact of the sea anemone Actinothoe sphyrodeta on Mytilus galloprovincialis mussel cultivation (Galicia, Spain). Biofouling. 2018 Nov;34(10):1138-1149. doi: 10.1080/08927014.2018.1547818. Epub 2019 Jan 30. PMID: 30698026.
- Fitridge I, Dempster T, Guenther J, de Nys R. The impact and control of biofouling in marine aquaculture: a review. Biofouling. 2012;28(7):649-69. doi: 10.1080/08927014.2012.700478. PMID: 22775076.
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.


 Save to Mendeley
Save to Mendeley
