Annals of Environmental Science and Toxicology
A critical review of biomarkers in toxicology and risk assessment of environmental pollutants
1Department of Biology, Polydisciplinary Faculty of Khouribga, University Sultan Moulay Slimane, Khouribga, Morocco
2Laboratory of Life Science and Earth (SVT), Regional Centers for the Professions of Education and Training (CRMEF), El Jadida, Morocco
Author and article information
Cite this as
Salama Y, Chennaoui M (2024) A critical review of biomarkers in toxicology and risk assessment of environmental pollutants. Ann Environ Sci Toxicol. 2024; 8(1): 6-18. Available from: 10.17352/aest.000075
Copyright License
© 2024 Salama Y, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Over the last 30 years, the study of biomarkers has experienced significant growth. Biomarkers serve as essential tools in monitoring toxicology and risk assessment of environmental pollutants by providing early and specific endpoints. This expansion in the field has enabled researchers to better understand the impact of various environmental pollutants on living organisms and ecosystems. By utilizing biomarkers, scientists can assess the potential risks posed by these pollutants and develop effective strategies for environmental protection and preservation. This article briefly reviews some of the aspects in an attempt to give an overall view of the field. Some of the new developments, particularly in relation to biomarkers of exposure and response, are mentioned.
Biomarkers represent crucial indicators or signaling events within biological systems or samples that demonstrate quantifiable alterations across multiple levels - encompassing molecular, biochemical, cellular, physiological, pathological, and behavioral domains - in reaction to external substances. The Biomarkers Definitions Working Group under the aegis of the National Institutes of Health (NIH) formally characterizes biomarkers as “objective measurements that serve as indicators of natural biological processes or pharmacological responses to therapeutic interventions” [1]. In the arena of toxicology, biomarkers are compartmentalized into three distinct categories delineated as markers of exposure, effect, and susceptibility. Through the examination of these biomarkers, deep-seated insights into the progression of injuries are gleaned, thus unveiling the intricate molecular pathways underpinning toxicity. These biomarkers play a pivotal role in enhancing diagnostic accuracy, offering precise prognoses, and devising effective treatment plans.
The identification of biomarkers for early chemical exposure may coincide with biomarkers heralding early disease detection, rendering valuable perspectives to preclude further chemical exposure and pave the way for tailored therapeutic interventions. By prioritizing individual patients over mere diseases, this method ensures that prevention outweighs treatment endeavors [2]. Biomarkers play a crucial role in the drug development process, spanning preclinical and clinical trials to ensure effective and safe assessment. These crucial indicators help in predicting, detecting, and monitoring any drug-induced toxicity across different stages, from preclinical studies to human clinical trials [3]. Developing sophisticated techniques and validating them for precise biomarker measurement, as well as interpreting the resulting data, are complex and time-consuming tasks that require significant dedication, effort, and intellectual expertise. Understanding the intricacies of drug metabolism is crucial in certain situations, as a drug’s metabolite can act as a valuable biomarker [4].
Historically, many medications have been developed alongside biomarker assays that guide their use. This trend is expected to continue in the field of drug discovery and development. By judiciously utilizing biomarkers, similar to evidence-based medicine principles, patients are more likely to benefit from targeted treatments while minimizing the risk of adverse effects. Conversely, the use of unreliable biomarkers can be as harmful to a patient as the use of ineffective drugs. Therefore, it is essential for biomarkers to undergo validation and evaluation by an accredited laboratory participating in a proficiency testing program. This rigorous process instills a high level of confidence among both healthcare providers and patients [5]. In the field of toxicology, biomarkers play a crucial role as predictive tools for toxicity testing and surveillance. These biomarkers must exhibit distinctive characteristics such as precision, sensitivity, authenticity, and biological or clinical significance. Additionally, their ease of use and efficiency are essential for enabling enhanced quantitative estimations of exposure and dosage. The importance of biomarkers is evident in their application in biomonitoring data, serving various purposes from assessing exposure to managing and evaluating risks.
The advancement and validation of novel laboratory techniques provide a foundation for valuable field methods. However, before implementing a new biomarker, certain fundamental information is required. This includes understanding dose-response relationships and identifying biological and environmental factors that can impact baseline response values. Notably, species differences can present significant challenges for toxicologists when working with biochemical or metabolic biomarkers.
Biomarkers play a vital role in various aspects of toxicology, particularly in relation to pesticides, metals, mycotoxins, and drugs. The field of veterinary toxicology also places significant emphasis on biomarkers, especially concerning plant toxins. Individuals in agricultural sectors, including farmers, pesticide applicators, and greenhouse workers, are directly exposed to pesticides, while their family members may face indirect exposure. The accurate assessment of pesticide residues, metabolites, and metals in urine through biomarkers is considered the most precise and reliable method for evaluating exposure in agricultural, industrial, and occupational safety and health contexts [6]. Recent studies suggest that exposure to specific pesticides, metals, and environmental contaminants during pregnancy or early life stages could potentially contribute to the development of various neurodegenerative disorders, such as Alzheimer’s, Parkinson’s, schizophrenia, Huntington’s, ALS, as well as metabolic, cardiovascular diseases, and cancer later in life. The identification of precise and trustworthy biomarkers is essential for the early detection of these conditions, enabling timely therapeutic interventions.
Mycotoxin-related toxicity, carcinogenesis, and a multitude of health issues are widespread among humans and animals worldwide. In developing nations, where regulatory standards may be less stringent, the repercussions can be profound, with reproductive and developmental impacts being especially harmful. Under these conditions, the early detection of biomarkers of exposure is essential, as it facilitates the mitigation of continued intake of contaminated food or feed, thereby safeguarding the well-being of humans and animals [7,8]. With the advanced understanding of systems biology, metabolomics, and various mathematical and computational/chemometric models, it has become feasible to uncover previously imperceptible biomarkers. These biomarkers hold promise in predicting how tissues respond to toxic substances, medications, and their metabolites. Moreover, they offer insights into the repercussions of tissue damage and regenerative mechanisms on overall tissue functionality. When juxtaposed with traditional biomarkers, imaging and chemometric biomarkers exhibit heightened sensitivity and furnish more exhaustive data. Not only are they capable of detecting minute quantities of chemical exposure (exposure biomarker), but they can also discern an early response from the tissue itself (endogenous response biomarker).
In the pursuit of advancing the identification of effective biomarkers, the amalgamation of innovative and pre-existing biomarkers through a multidisciplinary approach proves to be highly fruitful. Employing a multi-biomarker approach provides a wealth of information and enhances accuracy compared to relying solely on a single biomarker. The emergence of cutting-edge technologies, such as microRNAs (miRNAs), holds great promise as reliable and robust biomarkers for the early detection of various conditions, including diseases, birth defects, pathological changes, cancer, and toxicity. Due to their remarkable stability in biofluids like blood, there is a rapidly growing interest in utilizing miRNAs as diagnostic, prognostic, and predictive biomarkers. The outlook for the clinical application of miRNA discoveries is exceptionally optimistic, particularly in the field of molecular medicine [9]. The application of miRNAs is still relatively new. In the near future, the integration of pharmacological and toxicological targeting of miRNAs will likely become a standard practice in the development of advanced therapeutic approaches [10]. However, there is a pressing need for the creation of more groundbreaking biomarkers that exhibit exceptional sensitivity (leveraging biotechnology-based methods), require minimal sample quantities, and offer high-throughput screening capabilities.
The purpose of the study is to conduct a critical review of biomarkers in toxicology and risk assessment of environmental pollutants. The aim is to analyze and evaluate the current state of biomarkers in these fields, with a focus on their effectiveness and relevance in assessing the impact of environmental pollutants on human health. The novelty of this study lies in its comprehensive and in-depth analysis of biomarkers, providing valuable insights into their potential applications and limitations in toxicology and risk assessment. The contribution of this research is to enhance our understanding of the role of biomarkers in assessing environmental pollutants and to provide recommendations for future research and developments in this area.
Biomarker definitions and applications
In the realm of human biology, exposure to environmental chemicals such as those found in food, drinking water, and air can instigate a cascade of biological events within the body. This cascade represents a continuum from initial external exposure to subsequent physiological reactions. These reactions may signal the presence of the chemical, adverse health outcomes, or increased toxicity influenced by individual characteristics. The biological processes set in motion by chemical exposure have the potential to incite cellular, molecular, organ, or systemic responses, alongside a range of biochemical, physiological, and morphological changes [11]. The concept of biomarkers has been delineated by multiple researchers and scientific entities, with minor variations in their definitions. Generally, molecular biomarkers are typically categorized as indicators of exposure, effects, or susceptibility [12]. An exposure biomarker serves to signal prior interaction with a chemical; this interaction could involve an external substance, a resultant product from the interplay between a xenobiotic molecule and endogenous constituents, or a modification that alters the state of the target molecule. These biomarkers are typically quantified in bodily fluids or tissues. An effect biomarker denotes the presence (and degree) of a biological response following exposure to a chemical. This response could manifest as an endogenous constituent, a gauge of the system’s functional capacity, or a modified state recognized as impairment or disease. A susceptibility biomarker indicates an increased sensitivity to a chemical’s effects, which could emerge as either the presence or absence of an endogenous element or an aberrant functional response to an administered challenge.
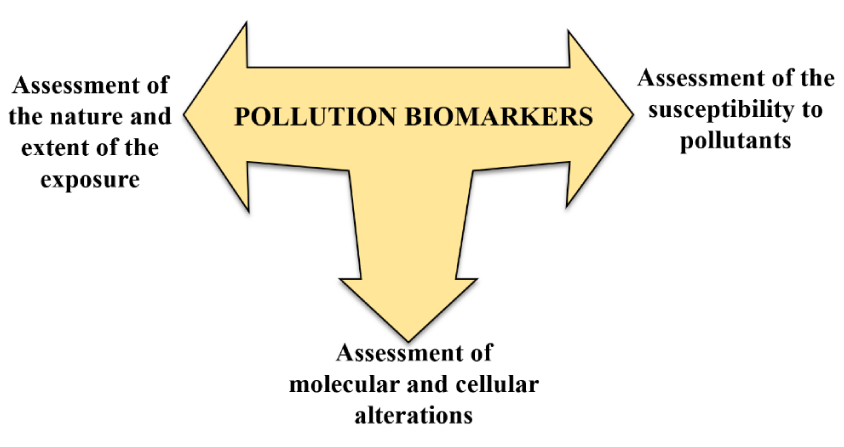
Biomarkers can be used to assess the nature and the extent of the exposure, to identify alterations occurring within an organism, and to assess the underlying susceptibility of an organism (Figure 1). They can help to increase the understanding of the processes by which a chemical is absorbed and transformed within an organism to determine alterations at the cellular and molecular levels leading to a toxic effect.
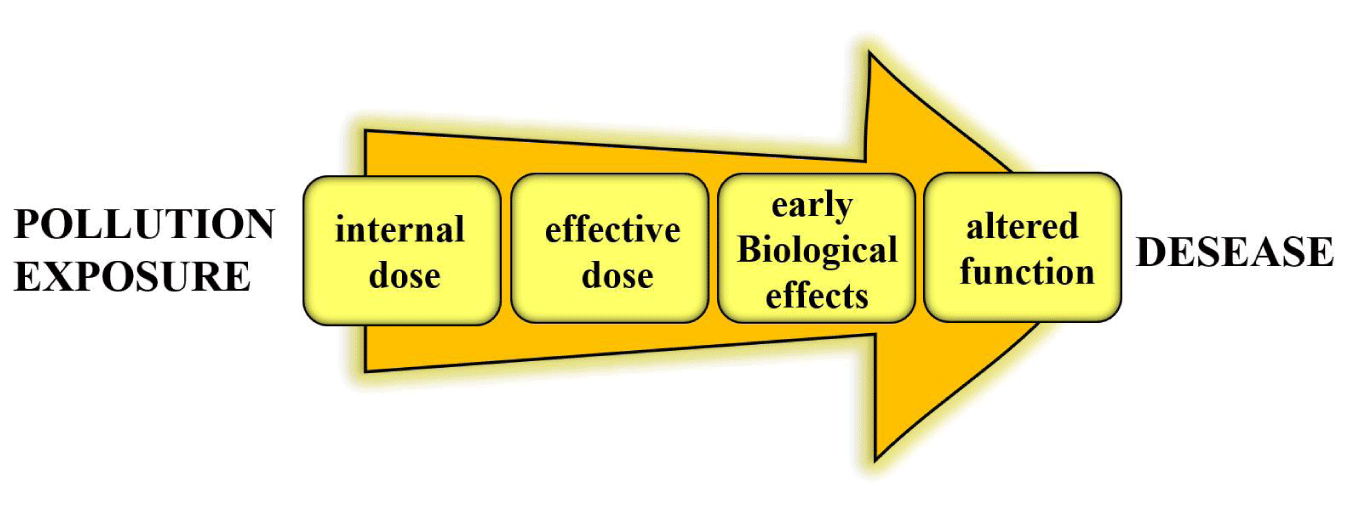
Therefore, depending on the specific biological response used as a biomarker and on the point on the continuum from exposure to pathology (Figure 2), where the measured biomarker comes from, biomarkers may be classified into biomarkers of exposure, biomarkers of effect, and susceptibility.
Various efforts have been made to define, develop, and validate an ideal biomarker [13]. Ideally, biomarkers have several common characteristics (Table 1). They are inexpensive, reliable, consistent, and easily measured, and their expression is altered under disease conditions.
It should be noted that even commonly used biomarkers are far from ideal but combinations of biomarkers for the same compound may give complementary information. For pesticides, the measurement of urinary biomarkers is a prevalent method employed to investigate exposure markers. About 30 chemicals or their metabolites are designated as urinary markers, with over 10 identified as serum markers for organochlorines, organophosphates, pyrethroids, carbamates, and other pesticides [14]. A recent study integrates urinary biomarker measurements from extensive and localized observational investigations to discern patterns that might not be readily discernible in examinations of individual studies [15].
In the field of risk assessment and chemical regulations, the United States Environmental Protection Agency (USEPA) performs risk assessments and establishes exposure limits for regulatory determinations. The agency endeavors to facilitate research aimed at discerning and assessing biomarkers that have the potential to forecast and/or predict pesticide exposure as well as the resultant health consequences stemming from such exposure [16], the US Environmental Protection Agency identifies biomarkers as presented in Table 2.
Biomarkers in toxicology and risk assessment
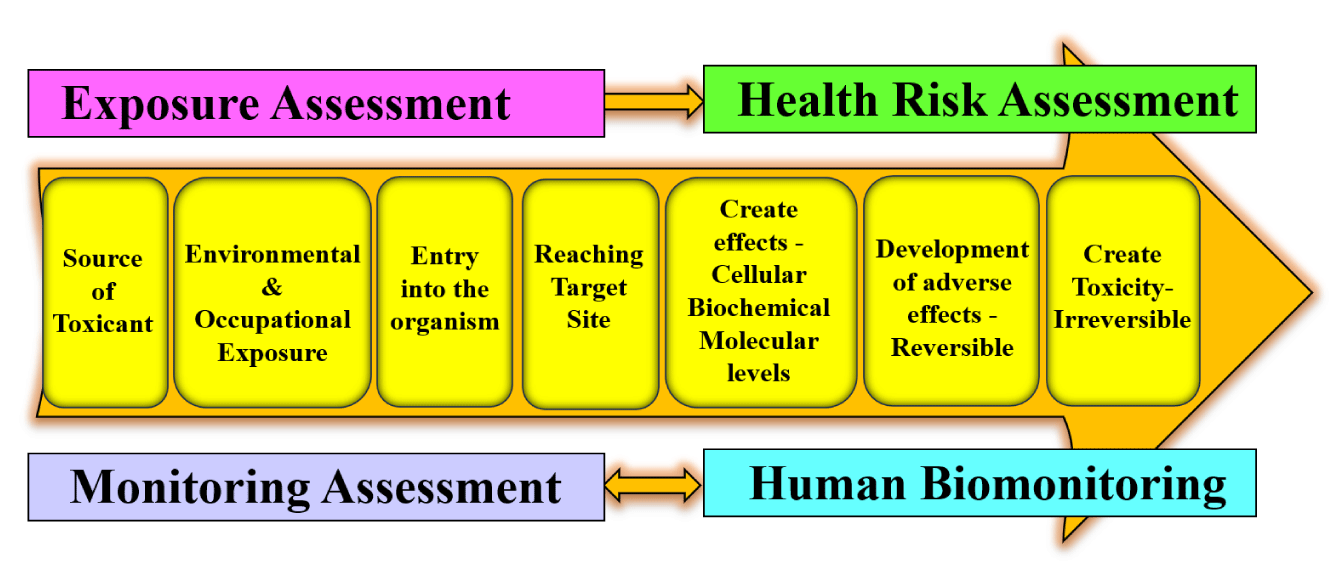
In the realm of environmental toxicology concerning human exposure to chemicals (whether natural or synthetic, excluding mainly physical and biological agents), toxicology focuses on understanding the harmful impacts of chemicals on individuals. Toxicologists carry out research and analysis to assess the health consequences of chemicals, aiming to determine their intended usage, safety levels, and potential risks associated with exposure, be it in natural settings or environmental contamination scenarios [17]. When any chemical is entered into an external environment, it passes through various phases creating toxic effects in an individual resulting in disease symptoms (Figure 3).
Risk assessment is the crucial process utilized by toxicologists to evaluate the potential for adverse health effects resulting from exposure to chemicals that can be found in various mediums like food, air, drinking water, soil, consumer products, the general environment, or the workplace. This thorough procedure typically entails assessing exposure and toxicological data collected from humans, animals, and other experimental systems [16]. Through this analysis, experts gauge the probability of harm or impacts that a chemical might present post-exposure, utilizing cause-effect and dose-response information while considering the biological underpinnings. Additionally, risk assessment involves scrutinizing various assumptions and uncertainties. To achieve this, the process encompasses four key steps: identifying hazards, evaluating dose-response relationships, assessing exposure levels, and characterizing risks. The outcomes of risk assessment are utilized to furnish a scientific foundation to aid risk managers in making decisions regarding risk management concerning the utilization of chemicals or their presence in the environment to safeguard human health [18]. Instances of such applications include establishing permissible limits for the formulation of regulatory standards for air quality, drinking water, or food safety. The decision-making process frequently encompasses other factors like societal values, technical feasibility, and economic considerations, which are outside the purview of this discussion.
The application of validated biomarkers can play a crucial role in understanding the sequence of events that unfold from the initial exposure to the subsequent response. This is pertinent in both clinical settings and epidemiological studies, where the aim is to delineate human exposure and leverage the ensuing effects to delineate the clinical manifestations that pave the way for disease development. In the domain of environmental toxicology and regulations, biomarkers aid in elucidating the clinical effects and diseases, with a specific emphasis on describing toxicological endpoints in human and animal experiments to facilitate a quantitative assessment of the cause-effect and dose-response relationships [12]. Certain biomarkers harnessed in molecular epidemiology have proven indispensable in toxicological research, especially in investigating the correlation between exposure and the emergence of diseases within exposed populations. Moreover, these biomarkers offer qualitative or quantitative cues on various stages of the toxicological process, encompassing cellular, tissue, or organism responses. They also illuminate individual susceptibility or host reactions, unraveling the mechanisms linking exposure to disease, particularly in scenarios where diseases manifest after a prolonged latent phase.
Molecular epidemiological data has the potential to offer valuable insights in establishing cause-and-effect relationships and understanding biological plausibility. By uncovering the associations between genetic variations and differences in susceptibility to the carcinogenic effects of a chemical, it becomes feasible to pinpoint potential carcinogenic hazards. When experimental evidence shows that a specific polymorphism changes the functional activity of the gene product in a manner that correlates with heightened susceptibility, or if the recognized phenotype of a genetic polymorphism sheds light on the fundamental mechanism of carcinogenesis for the agent being assessed, then such data can play a crucial role in making causal inferences [19].
Although biomarkers used in epidemiology are measures of the continuum of events that lead to certain toxicological endpoints, and the continuum of cellular or molecular responses to chemical exposures may provide linkages to mechanisms of biological response, they may not be by themselves the endpoints used in risk assessment and regulations development [20]. One critical aspect of risk assessment is the quantitative assessment using hazard identification and dose-response data. Whereas one of the purposes of biomarkers in environmental and public health is to identify the effect of concern and the highly exposed individuals or groups, so that the identified risk can be predicted and disease prevented, biomarkers are rarely the basis of exposure-response relationships, such as the case of urinary cotinine, except for example such as blood lead, blood mercury, and red blood cell (RBC) cholinesterase (AChE) inhibition.
Epidemiological studies in human populations are more likely to be relevant for human risk assessment when the data are of high quality, as opposed to findings from animal toxicity studies. However, various confounding factors such as concurrent exposures to other chemicals, differences in individual lifestyles, challenges in accurately reconstructing exposures, and the small sizes of most cohorts can hinder the ability of epidemiological studies to unambiguously determine risks to humans at low levels of exposure [21]. Precision in estimating risks to human health resulting from low-level exposure to potentially hazardous chemicals in the environment demands a thorough understanding of the dose-response relationship across a wide spectrum of exposures in animal species. This includes dose-response curves that identify risks in animals at low response levels, though such data are frequently lacking.
In order to be valuable for risk assessment, biomarkers need to not only determine the presence or absence of an exposure or effect but also aid in quantifying the exposure and dose-response in some capacity. The extrapolation of human, animal, or other experimental data to real-life human scenarios is a conventional approach to assessing potential health risks from chemical exposures. Molecular epidemiology offers the advantage of directly applying human data to human situations when good-quality data is available, thereby obviating the necessity for interspecies extrapolation. However, causal relationships and quantitative dose-response data may not always be accessible.
Types and roles of biomarkers: A comprehensive
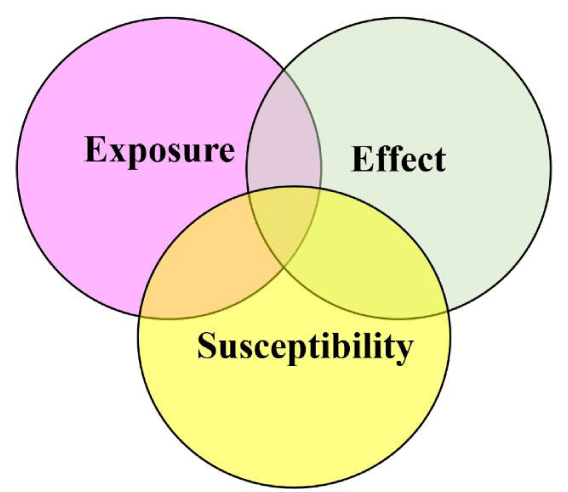
A biomarker reflects an event or a sequence of events that occur somewhere in the causal chain between an exposure to a hazardous factor and a related adverse event effect. Theoretically, these events can be separated into those indicating “internal dose,” “effective dose,” etc., but some of these terms themselves are difficult to define, and a detailed classification of biomarkers may not necessarily add to the understanding of their properties. nevertheless, three specific types of biomarkers are usually identified (Figure 4).
Although some biomarkers may belong to more than one class, they are often separated into biomarkers of exposure, biomarkers of effect, and biomarkers of susceptibility. The different types of biomarkers, categories, biological matrices, and outcomes are given in Table 3.
Biomarkers of susceptibility
Biomarkers of susceptibility represent a complex and challenging aspect of the risk assessment process, often characterized by a lack of comprehensive understanding. These biomarkers primarily focus on factors that influence the absorption, distribution, metabolism, and excretion of chemicals, as well as their subcellular biological effects. They are heavily influenced by metabolic enzymes, which play a pivotal role in modulating an individual’s susceptibility to disease. Genetic variations in the expression of these metabolic enzymes can significantly contribute to inter-individual differences in susceptibility. Such genetic differences have the potential to impact the progression and severity of effects caused by exposure to various chemicals. For instance, variations in enzymatic genotypes and phenotypes, such as those observed in glutathione-S-transferase M, a phase III conjugation enzyme involved in detoxifying electrophilic compounds, may play a crucial role in determining an individual’s susceptibility to certain health outcomes. Ongoing research focuses on evaluating the genotype and phenotype of metabolic enzymes across diverse populations to identify potential associations between exogenous exposure and specific metabolic genotype subsets. This analysis aims to enhance our understanding of how genetic factors can influence an individual’s response to chemical exposure and subsequent health effects.
Point mutations or small alterations in gene sequences have the potential to result in heritable gene variants. These gene variants, while not themselves indicative of disease, serve as the basis for individual differences within populations. When the frequency of a gene variant exceeds 1% within a population, it is termed a genetic polymorphism, with particular emphasis placed on those variants that exhibit functional consequences. The enzymes responsible for the activation and detoxification of foreign substances, known as xenobiotics, can be categorized into two groups. The first group consists of phase I enzymes, prominently represented by the cytochrome P450 superfamily of mixed-function oxidase enzymes. The second group comprises phase II enzymes, which facilitate the conjugation of oxidized substrates with diverse moieties such as glucuronic acid, glutathione, and sulfate. Fluctuations in biomarker levels are likely connected to polymorphisms present in Phase I and Phase II enzymes involved in biotransformation and DNA repair processes. Genes involved in xenobiotic metabolism, including the cytochrome P450 enzymes CYP1A1, CYP1A2, CYP2A6, CYP2D6, CYP2E1, N-acetyl-transferase 1 and 2 (NAT1 and NAT2), and Glutathione S-Transferase (GST) and theta (GSTM1 and GSTT1), exhibit polymorphisms within human populations. Studies have indicated ethnic disparities in the transcription and translation patterns of these enzymes across diverse population groups. Among the extensively researched cytochrome P450 variants, CYP1A stands out for its polymorphic nature. CYP1A2, for instance, plays a key role in the N-oxidation of various aromatic and heterocyclic amines into DNA-reactive species. Notably, alterations in CYP1A2 function have been linked to an elevated susceptibility to colorectal and bladder cancers in humans.
Various biomarkers have been extensively researched and elucidated. The acetylator biomarkers constitute another category predicated on metabolic susceptibility genes relevant to exposures to aromatic and heterocyclic amines. N-acetyl-transferases are under the auspices of two distinct genes, situated on chromosome 8 in humans, denoted as NAT1 and NAT2. Polymorphisms in NAT2 manifest as point mutations in the coding regions of this gene lacking introns, culminating in differing metabolic phenotypes. Homozygosity for the slow acetylator gene characterizes slow acetylators, a genetic predisposition identified in diverse populations including Canadian Eskimos (5%), Japanese (10% - 20%), Caucasians (50% - 60%), and Northern Africans (90%). It is noted that although studies that measure cell functions, enzymes, or metabolites that are thought to be the basis of susceptibility may provide evidence that supports biological plausibility when data on genetic susceptibility originate from multiple comparisons that arise from subgroup analyses, this can generate false-positive results and inconsistencies across studies.
Biomarkers of exposure
Biomarkers of exposure are typically evaluated through direct analysis of the presence of the specific chemical substance, its metabolites, or its constituents in bodily tissues or fluids of individuals, such as hair, urine, or blood. The detection of a biomarker merely indicates that an exposure has taken place, without necessarily implying any biological significance such as the presence of disease or adverse effects; in certain instances, it might suggest that an individual is potentially susceptible to toxicity. Moreover, the identified biomarker may not necessarily represent the specific toxicological endpoint of concern for risk assessment. Biomarkers of exposure might also encompass the identification of macromolecular reaction products like hemoglobin adducts, which do not directly contribute to adverse outcomes. The toxicological implications associated with these biomarkers rely on various factors, including but not limited to the study’s nature, purpose, and methodology; the assessment tools utilized; the extent and type of exposure; as well as the biological, chemical, physical, and toxic characteristics of the compound in question. Additional crucial factors contributing to the toxicological interpretation of biomarkers comprise the biological, biochemical, physiological, pathological, and toxicological endpoints of interest, along with the sensitivity of the organism’s response to the exposure [22].
Among the categories of biomarkers, biomarkers of exposure have garnered substantial attention within the scientific community. Watson and Mutti [23] intricately elucidated the significance of biomarkers and delineated four distinct categories predicated on biological half-life. These categories encompass biomarkers with varying duration, ranging from very short, such as phenol for benzene exposure; to short, exemplified by 2,5-hexanedione for hexane exposure; to long, as observed in heavy metals such as lead, mercury, and cadmium in blood, as well as DNA and hemoglobin adducts for electrophilic compounds or metabolites; and finally, to very long, typified by heavy metals in bone. Exposure biomarkers offer invaluable insights by quantifying the presence of chemicals in biological matrices. For instance, lead and dioxins in blood, mercury in hair, and cadmium in urine serve as indicators of chemical presence within the body. However, it is crucial to acknowledge that these biomarkers alone may not comprehensively signify the toxicological ramifications unless complemented with data on dose-response relationships concerning toxicity, risk evaluation, and established exposure or regulatory thresholds.
There exists a functional connection between the external dose (inhalation, ingestion, dermal absorption), the internal dose (as detected in body fluids or tissues), and the related biological impact. These factors play crucial roles in risk assessment and risk management. While the dose-response patterns of biomarkers may not mirror the intricate curves observed in comprehensive responses, the monitoring and pharmacodynamic modeling of alterations in these molecular and biochemical indicators remain invaluable for deriving an upper-limit risk evaluation at minimal exposure levels. Given that the association between toxic reactions and biomarker alteration extent is unlikely to be uniform across all doses, estimating risks at low concentrations may not significantly enhance precision via extrapolation using these biomarkers.
Biomarkers of effect
A marker of effect may represent an endogenous component, a measure of functional capacity, or another indicator of the body or organ system’s state or balance influenced by exposure. These effect markers typically serve as preclinical signs of abnormalities. Biomarkers can be either specific or nonspecific. Specific biomarkers prove valuable as they demonstrate the biological impact of a particular exposure, potentially aiding in preventive measures. Conversely, nonspecific biomarkers do not pinpoint a single cause but instead reveal the overall integrated effect resulting from a combination of exposures.
As Harris, et al. (7) discuss, biomarkers play a critical role in assessing the genotoxic effects of environmental chemicals. Methods used to evaluate genotoxicity in humans can also be applicable in animal studies, allowing for comparisons to target tissues inaccessible in human research. By tracing causal chains, these studies can yield new insights into disease development. Biomarkers thus offer a shared platform for collaboration between toxicologists and epidemiologists. It is not surprising that this field of research has garnered significant interest, particularly concerning cancer. Biomarkers of effect have not been as widely employed as exposure markers. Examples include inhibition of RBC cholinesterase (ChE) activity and development of methemoglobinemia [24]. Reversibility of the effect may occur upon cessation of exposure, evidenced by a subsequent increase in RBC ChE activity and a decrease in blood methemoglobin levels. Given the continuum of biomarker events, it is often challenging to clearly differentiate between effective dose markers, early effect markers, and markers of adverse effects. Various degrees of RBC ChE activity inhibition and blood methemoglobin levels indicate varying levels of severity of effects, which can ultimately result in fatality at extremely high effect levels [25].
Cancer-related biomarkers: detecting and understanding the disease
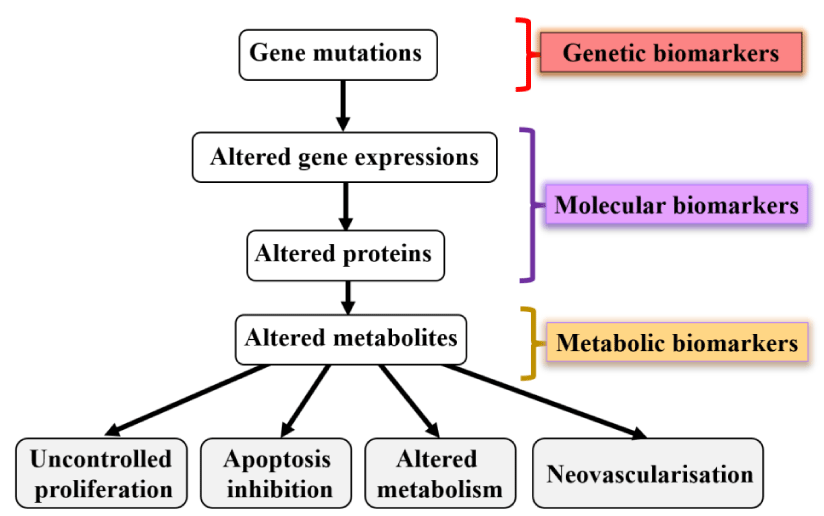
In the area of cancer research, molecular and cellular biomarkers serve as essential tools for the assessment of cancer susceptibility [26]. The examination of biomarkers pertaining to exposure or bioactive dose includes analysis of DNA adducts, as well as surrogate indicators like protein adducts and metabolites present in urinary excretion. Notable biomarkers associated with biological effects encompass gene mutations, chromosomal aberrations, and micronucleus formations (Figure 5).
The substances created when a carcinogen interacts with DNA or protein are known as adducts, marking a crucial stage in the development of cancer. Genotoxic carcinogens require the study of biomarkers, including measuring the carcinogen or its active metabolites in bodily fluids, determining the presence of adducts, or quantifying specific biological effects of the adducts, such as mutations or cancer [27]. Reactive molecules generate DNA adducts through the metabolic activation of carcinogens’ ultimate forms, leading to covalent interactions with cellular DNA, ultimately forming adducts. Adducts serve as distinctive biomarkers, offering a method to gauge human exposure to chemical carcinogens and comprehend the biologically effective dose at a significant carcinogen target site. If DNA replication occurs without or before repairs are made, mutations may occur. Nonetheless, the molecular event itself does not directly apply to risk assessment. The measurements merely symbolize a qualitative link between the organism’s biomarker and the risk of evident toxicity, or an early event. Conversely, even in the absence of indications of an increased response in biomarkers, there might still be a degree of escalation in the biomarker and hence in the risk level, which cannot be completely assessed based solely on the measurements owing to insufficient data on quantitative measurements for low-level exposures [28].
DNA adducts have proven to be invaluable tools for studying human cancers. Among the various applications, the two have been particularly well-established and validated. Studies have shown a clear link between tobacco smoking and lung cancer, as well as between Aflatoxin (AF) exposure and liver cancer [29]. Tobacco smoke is known to be a major contributor to the development of lung and other cancers. It contains a range of carcinogens, including aromatic amines, Polycyclic Aromatic Hydrocarbons (PAHs), and tobacco-specific nitrosamines like the lung-specific carcinogen nicotine-derived nitrosamine ketone NNK. These carcinogens undergo metabolic activation to form reactive species that lead to the formation of specific DNA adducts.
Studies have revealed that smokers, when compared to nonsmokers or ex-smokers, exhibit several key differences in their DNA adduct profiles [30]. These include significantly elevated levels of aromatic and/or hydrophobic adducts, a linear relationship between DNA adducts levels and total smoking exposure, higher levels of bulky, hydrophobic DNA adducts in lung tissue, a positive correlation between bulky adduct levels and the activity of CYP1A1 (Aryl Hydrocarbon Hydroxylase (AHH)), a significant correlation between pulmonary microsomal AHH activity and the levels of benzopyrene-DNA adducts, and increased PAH-DNA adducts in combined DNA samples from lymphocytes and monocytes [31]. Both human and animal studies have offered data on the validation of these carcinogen adducts as biomarkers. In consecutive samples from heavy smokers, the PAH-DNA adducts and 4-aminobiphenyl-hemoglobin (4-ABP-Hb) adducts in peripheral blood showed a significant decline of 50% - 75% once smoking was discontinued. The PAH-DNA adducts in leukocytes were found to have an estimated half-life of 9 weeks - 13 weeks, while the half-life of 4-ABP-Hb adducts was estimated to be 7 weeks - 9 weeks [32].
Aflatoxin B1 is a food contaminant that is linked to the induction of liver cancer in Asia and Africa, especially among carriers of the hepatitis B virus [33]. Aflatoxins are significant components of mycotoxins produced by specific molds in crops that are inadequately stored in warm and humid regions and are present as contaminants in rice, peanuts, maize, and various cereals and animal feed. In rats, a linear relationship between the dose of AFB1 and the excretion of the primary nucleic acid adduct, AFB1-N7-Guanine (AFB-N7-Gua), was observed within the initial 24-hour period following a single exposure to AFB1; this correlation was not observed with other oxidative metabolites. Epidemiological studies in Taiwan and China have revealed a link between carcinogen-specific biomarkers like serum AF-albumin adducts and the risk of liver cancer [34]. A nested case-control study in Taiwan involved a cohort of 8068 men monitored for 3 years, showing an association among men under the age of 52, but no association between AFB1-albumin adducts level and hepatitis B antigen (HBsAg) carrier status. Another study is a prospective nested case-control study conducted in Qidong, China, involving 804 healthy HBsAg-positive individuals aged 30 - 65, with a follow-up period of 4 years.
Proteins used for adducts determination possess a prolonged lifespan and robust stability. It is worth mentioning that protein adducts are also detected at baseline levels in individuals who have not been exposed to certain substances, such as acrylamide, ethylene oxide, acrylonitrile, and styrene oxide, as well as certain amino acid adducts like 2-carboxami-doethyl-valine, 2-hydroxyethyl-valine, 2-cyanoethyl-valine, and 2-hydroxy-2-phenylethyl-valine [35]. Numerous chemical carcinogens exhibit binding capabilities to both DNA and blood proteins in a manner that follows similar dose-response patterns. Consequently, the formation of carcinogen-protein adducts serves as a useful proxy for DNA adducts. Within this context, Hemoglobin (Hb) and serum Albumin (Alb) emerge as preferred protein targets. Hemoglobin boasts an approximate lifespan of 60 days in rodents and 120 days in humans, while serum albumin has a half-life of 23 days in humans. Due to the innate stability of protein adducts and their resistance to active repair mechanisms, they present a significantly more precise tool for dosimetry when compared to DNA adducts.
Formation of Hb or serum albumin adducts has been reported in experimental animals and humans for carcinogens, including AFB1, aromatic amines, BaP, benzene, dimethylnitrosamine, ethylene oxide, 2-amino-3-methylimidazo[4,5-f]quinoline, methylmethane sulfonate, NNK, propylene oxide, styrene, and workplace and medicinal PAHs [36]. The carcinogen-Hb adducts formed by the potent urinary bladder carcinogen 4-ABP, and 4-ABP-Hb adduct, are closely associated with three major risk factors for bladder cancer: cigarette smoking, the type of tobacco smoked, and acetylator phenotype.
Haschek, et al. [37] conducted a framework analysis to examine the mode of action of genotoxic chemicals and challenge the prevalent notion that cancer risk follows a linear trend at extremely low doses. Their study involved utilizing biomarkers of exposure to investigate variations in DNA damage and repair across different species, tissues, and cell types. This comprehensive approach incorporated factors such as exposure levels, metabolic activation, and detoxification processes, along with data on biomarkers of effect to gauge responses to gene and chromosomal mutations resulting from exposure. The analysis revealed that biomarkers of exposure demonstrate a linear relationship at low doses, except when identical adducts are formed endogenously. Typically, sublinear responses are observed at doses with impaired detoxication or DNA repair, leading to a disproportionate increase in adducts per unit dose at high concentrations. Conversely, supralinear responses are linked to the saturation of metabolic activation, resulting in fewer electrophiles formed per unit dose of elevated exposures. Below these nonlinear processes, DNA adducts are expected to show linear associations with administered doses. Examples have established dose-response relationships for DNA and protein adducts following exposure protocols akin to those used in animal carcinogenicity bioassays. The molecular dose encompasses processes like absorption, distribution, metabolism, detoxification, and DNA repair. The intricate balance between electrophile formation and stability, adduct stability and repairs, protein lifespan, and cell death dictates the distribution, shape, and temporal evolution of the measured molecular dose. While biomarkers of exposure can be extrapolated to zero, biomarkers of effect can only be interpolated to the spontaneous or background mutation rate. This distinction arises because high exposure levels drive mutagenesis through DNA damage caused by chemical exposure, while very low exposures spur endogenous DNA damage. The dose-response curves of biomarkers of effect offer valuable insights for estimating cancer risk resulting from multiple mutations, although data on mutagenesis at low doses are scarce for most chemicals.
Many cancers carry the TP53 mutation. The tumor suppressor gene p53 has been utilized as a biomarker for molecular carcinogenesis, molecular epidemiology, and anticipated cancer risk. It is the most frequently mutated gene identified in human cancers, with a prevalence of 44% in colorectal cancer and 38% in lung cancer [38]. The quantity and nature of mutations in this gene arise in specific hotspots that differ according to tumor types, as variations in mutation profiles among tumors have been observed due to distinct etiologies for the respective tumor types. A definitive circumstantial association between AF exposure and the alterations identified in p53 in liver tumors has been established in studies investigating the connection between AF exposure and the onset of human Hepatocellular Carcinoma (HCC) in China and Southern Africa [39]. Three separate investigations into p53 mutations in HCCs detected in populations exposed to elevated levels of dietary AF revealed a high prevalence of G to T transversions, with clustering predominantly at codon 249. However, this specific codon 249 mutation was not found in analyses of p53 mutations in HCCs from Japan and other regions with minimal AF exposure [40].
Adducts play a limited role in assessing the carcinogenic risk, except for AF1, where the identification of adducts has aided in analyzing the risk of cancer in humans. Specifically, a correlation was observed between levels of urinary aflatoxin B1-N7-guanine adduct and HCC in a study conducted in Shanghai, China [41]. However, no significant link was established between dietary intake of AF1 and cancer risk based on questionnaire data. While in experimental systems, it is feasible to establish a direct relationship between the formation of adducts and tumorigenesis, as well as to establish connections between adduct levels and tumor development [42], such analyses have not yet been carried out in human subjects.
Biomonitoring has aided in the recent discovery of human exposure to acrylamide through diet, resulting in the detection of acrylamide-globin adducts. This further enhances understanding of the presence of this carcinogen in food items rich in carbohydrates and asparagine, which are cooked at high temperatures (e.g., baking and frying), like fried potatoes [23].
Biomarkers of susceptibility
Biomarkers of susceptibility represent a complex challenge in risk assessment due to limited understanding and intricate integration into the evaluation process. These biomarkers primarily focus on factors influencing the kinetics (absorption, distribution, metabolism, and excretion) and dynamics (subcellular biological effects) of chemical compounds, heavily influenced by metabolic enzymes [14]. Genetic variations in the expression of these enzymes play a significant role in interindividual susceptibility to diseases, potentially altering the progression of chemical effects. Enzymatic genotypes and phenotypes, such as those associated with glutathione-S-transferase M, a phase III conjugation enzyme involved in detoxification, may significantly impact susceptibility. Ongoing research across diverse populations aims to determine the genotype and phenotype variability of metabolic enzymes, seeking to establish connections between exogenous exposure and specific metabolic genotype subsets leading to potential health effects.
Point mutations or small sequence alterations can result in inherited gene variants, as discussed by Anna and Monika [43]. These gene variants are not typically associated with disease but instead contribute to interindividual variability. When a gene variant is present in at least 1% of the population, it is referred to as a genetic polymorphism, with those exhibiting functional effects being of particular significance. Enzymes responsible for the activation and detoxification of xenobiotics are categorized into two main groups: phase I enzymes, primarily the cytochrome P450 mixed function oxidase enzyme superfamily, and phase II enzymes, which conjugate oxidized substrates with various moieties like glucuronic acid, glutathione, and sulfate.
Variations in biomarker levels are often linked to polymorphisms in the Phase I and Phase II biotransformation enzymes and DNA repair enzymes. Genes involved in xenobiotic metabolism, such as cytochrome P450 enzymes like CYP1A1, CYP1A2, CYP2A6, CYP2D6, CYP2E1, N-acetyltransferases 1 and 2 (NAT1 and NAT2), and glutathione S-transferase (GST) and theta (GSTM1 and GSTT1), exhibit polymorphisms in human populations. Studies on the genotypes of human cytochrome P450 enzymes across diverse populations have revealed ethnic differences in the transcription and translation of these enzymes. One of the extensively studied cytochrome P450s concerning polymorphism is CYP1A. CYP1A2, for instance, plays a vital role in catalyzing the N-oxidation of various aromatic and heterocyclic amines into DNA-reactive species. Research has shown an association between CYP1A2 and an increased risk of human colorectal and bladder cancer [44].
Gene variants for which meta- or pool analyses have suggested significant effects on cancer risk include the following: CYP1A1, implicated in lung cancer in both smokers and non-smokers; CYP1A1, associated with lung cancer in smokers and non-smokers as well as esophageal cancer; CYP1A1/STM1, linked to lung cancer; GSTM1, identified in head and neck cancer and acute leukemia; CPY1A1 wild type/GSTM/GSTT non-null, showing risk reduction for lung cancer in non-smokers; GSTT1, associated with lung cancer in non-smokers, breast cancer, and acute leukemia; GSTM1/GSTP/GSTT1 null, related to head and neck cancer; XRCC1 Arg280His, implicated in various cancer types; XRCC1 Arg194Trp, providing a protective effect against all cancers; XPD Lys751Gln, associated with lung cancer; and XPD Asp312Asn, linked to lung cancer [45]. Studies on genetic susceptibility often lack exposure information, and it is expected that individuals may carry variants impacting DNA damage response pathways, potentially leading to significant consequences even with moderate pathway inefficiencies.
It has been observed that while investigations evaluating cell functions, enzymes, or metabolites believed to underlie susceptibility may offer support for biological plausibility, the presence of genetic susceptibility data stemming from numerous subgroup analyses can lead to the generation of false-positive outcomes and study inconsistencies [46].
Molecular mechanisms and risk assessment
“Omics” embodies a pioneering evolution in biomarker methodology, leveraging cutting-edge high-throughput techniques that integrate advanced robotic and instrumental procedures, image analysis, and bioinformatics. These methods are utilized to effectively manage and analyze the extensive amount of data produced by these technologies [32]. This approach holds the potential to amplify our understanding of the molecular mechanisms underlying carcinogenesis, aiming to strengthen environmental health risk assessment and guide decision-making for impactful environmental health policies. According to Gupta [25], there are various classes of biomarkers known as “omics” biomarkers that play a significant role in the mode of action-based risk assessment. These classes include genomic biomarkers (such as genotyping, gene expression, and epigenomic biomarkers), proteomic biomarkers, and metabolomic/metabonomic-based biomarkers.
Proteomic biomarkers play a pivotal role as indicators of gene expression, enabling researchers to delve into shifts in protein profiles. By scrutinizing these profiles, scientists gain insights into how genes are translated into proteins within cells and bodily fluids such as blood or urine. The expression of proteins mirrors the functional reactions of genes that are either boosted or suppressed in response to exposure to various chemicals. The analysis of proteomic data offers invaluable perspectives on how cells react to different chemicals, especially in distinct cell populations. It also aids in pinpointing vulnerability markers based on the expression of protective proteins like stress proteins or metallothionein. Moreover, proteomic biomarkers serve as tools for gauging the toxicity of chemicals on target organs, evident through urinary protein excretion patterns [47]. The alterations in proteomic profiles and their correlation with specific chemicals furnish a holistic understanding of how toxic substances impact individual cells and populations. This information is derived from experimental systems and takes into account factors like the type of chemical, the mixture of chemicals, and the duration and intensity of exposure. These proteomic responses reflect the mechanisms of toxicity, such as Reactive Oxygen Species (ROS), by indicating the cellular pathways that respond to chemical exposure. When proteins undergo oxidation, known as proteotoxicity and indicative of ROS, it triggers the upregulation of major stress protein classes as an initial protective response by the cells. By examining the nature of the proteomic response, we can obtain easily understandable information for assessing the potential health risks associated with persistent exposure. If the protective stress protein responses are weakened and correlated with increased cellular toxicity (e.g. proteinuria), it can suggest the limits of protection. Although the application of this information to human health or epidemiological studies has been limited, it has provided valuable insights into identifying populations at specific risk. Genomic biomarkers play a critical role in evaluating cellular responses to chemical exposures at the genetic level [48]. They offer essential indicators of alterations in specific genotypes, such as d-aminolevulinic acid dehydratase (ALAD), as well as the up-or-down-regulation of particular genes like stress proteins and metallothionein. Additionally, these biomarkers involve epigenetic changes, such as modifications in DNA base methylation. By studying genomic expression data, researchers can uncover valuable insights into susceptibility biomarkers linked to genetic variations that can impact an individual’s sensitivity to harmful substances. Moreover, epigenomic strategies enable the exploration of how changes in DNA activity regulation may affect susceptibility and the likelihood of developing various adverse health conditions, including cancer.
Research on metabolomics and metabonomic biomarkers utilizes analytical methods to trace the disturbances caused by chemicals in metabolic pathways. These disturbances manifest through the production of metabolic substrates or products within said pathways [25]. The primary emphasis is on quantifying metabolites stemming from intermediary metabolism. By noting the rise in the presence or elimination of metabolic precursors or products in biological samples post-chemical exposures, valuable understandings can be acquired regarding the interplay between these agents and vulnerable metabolic pathways. Additionally, this data illuminates potential adverse biological impacts and the importance of disruptions occurring in these pathways. For instance, exposure to lead may induce significant changes in the heme biosynthetic pathway as a compelling demonstration. Specific metabolic pathways and reactive intermediates have been implicated in the mechanism of action for bladder cancer induced by Disinfection By-Products (DBPs) such as trihalomethanes (THMs - bromoform, chloroform, bromodichloromethane, chlorodibromomethane). The metabolism is mediated by enzymes from both the CYP450 and Glutathione S-Transferase (GST) families, both of which have shown polymorphisms and interethnic differences in CYP2E1 polymorphism [49].
Integration with environmental regulations
In the realm of environmental chemical regulations, toxicologists are pivotal in executing risk assessments to aid decision-making on risk management. These determinations frequently culminate in the development of guidance levels or regulatory benchmarks. Throughout this undertaking, it is imperative for toxicologists to delineate a quantifiable basis for overseeing chemicals that present health hazards. This basis must establish a qualitative or quantitative correlation between detrimental health outcomes and exposure to said chemicals in the ambient environment.
Biomarkers have long served as surrogate measures of biological impact in both laboratory and field studies, as well as in the biomonitoring of the general population. Despite their widespread utility, the integration of biomarkers into the quantitative risk assessment of environmental chemicals for regulatory purposes has been somewhat constrained. Molecular parameters have shown limitations as reliable predictors of actual risk and may fail to delineate the dose-response relationship from a mechanistic standpoint. Only biomarkers capable of identifying critical health effect endpoints and exhibiting changes interpretable in a dose-response manner are considered suitable for such endeavors [50].
While biomarkers have furnished valuable insights into specific exposures concerning cancer etiology within populations, the majority of these biomarkers, except for a select few, lack adequate validations and characterization for the precise quantitative estimation of cancer risk. Consequently, data on cause-effect relationships and dose-response correlations often remain inadequately documented. Human surveillance studies on disease burdens have shed light on the cumulative impacts stemming from various risk factors, although they fall short of offering a comprehensive attribution of individual risk factors to specific diseases [51]. Such attribution stands as a critical prerequisite for effective regulatory measures. There exists a pressing need to establish a measurable foundation for regulating causative agents that establish a definitive link between disease outcomes and environmental sources. Of particular importance is the acquisition of quantitative dose-response data that can be leveraged for comprehensive risk assessments.
Environmental programs are charged with the task of conducting research and assessing the toxic properties of environmental chemicals. They are further tasked with conducting risk assessments and offering assistance in formulating chemical regulations. As previously indicated, biomarkers assume a critical function in toxicology and risk assessment endeavors. Therefore, it is pertinent to examine the integration of particular biomarkers within the precise chemical regulations tied to these programs. At the federal level, drinking water contaminants are regulated under the National Primary Drinking Water Regulations (NPDWRs) [52]. The NPDWRs, also known as primary standards, are legally enforceable and are applicable to public water systems. Their purpose is to safeguard public health by limiting the concentrations of contaminants in drinking water. Specifically, the regulations address various aspects including chemicals (both organic and inorganic), maximum contaminant levels (MCLs), and maximum contaminant level goals (MCLGs).
MCLGs represent the level of a contaminant in drinking water that poses no known or expected health risks. These goals provide an additional margin of safety and are not binding regulations, but rather serve as important public health objectives. On the other hand, MCLs denote the allowable highest concentration of a contaminant in drinking water. These MCLs are set as close to MCLGs as feasible using the best available treatment technology and taking cost into consideration. MCLs are enforceable standards [53]. In regards to drinking water and chemical regulations, it is noteworthy that the State of California has set forth Maximum Contaminant Levels (MCLs) for chemical pollutants. These MCLs may align with or surpass federal standards. The Public Health Goal (PHG) program conducts toxicological assessments and risk evaluations to determine the permissible levels of contaminants in drinking water. These guidelines ensure that the daily consumption of water does not pose notable health risks over a person’s lifetime. The PHGs are derived from observed cancer or noncancer effects and mirror the federal Maximum Contaminant Level Goals (MCLGs) for drinking water. Per the California Safe Drinking Water Act of 1996, the Office of Environmental Health Hazard Assessment (OEHHA) conducts risk assessments and issues PHGs to prioritize public health considerations without factoring in costs. MCLs are set as close to the PHGs as feasible, considering factors such as cost and technical feasibility.
Out of the 82 Public Health Goals (PHGs) that have been developed, 40 are focused on systemic effects, while the remaining 42 are centered on carcinogenic effects. Biomarkers of effect for systemic effects include nitrate and selenium. Conversely, for carcinogenicity, cancer potency is established for each chemical identified as a carcinogen, and a PHG is set at the 1026 risk level. Carcinogenicity determinations are predominantly derived from animal studies rather than human epidemiologic studies, all revolving around the tumor or cancer incidence data. Cancer risk assessment hinges on the study showcasing the highest tumor incidence. The current guidelines for cancer recommend the application of linear extrapolation for genotoxic carcinogens [54,55]. In cases involving chemicals exhibiting both systemic effects and carcinogenicity endpoints, a health-protective concentration is determined for each of these outcomes. The concentration that offers greater protection to human health is subsequently chosen as the Primary Health Goal (PHG). For example, benzene, which is hepatotoxic and carcinogenic, sets the PHG based on its carcinogenicity. Pertaining to carcinogens, current information on adducts and genotoxicity, encompassing chromosomal effects (as earlier mentioned biomarkers of exposure and effect), serve as additional mechanistic data for assessing the weight of evidence and discerning whether a substance is a genotoxic or non-genotoxic carcinogen. Nevertheless, these details are not directly used for risk assessment.
The primary focus of P65 toxicology revolves around the compilation of chemicals known for their potential to induce carcinogenesis or reproductive toxicity. According to statutory requirements, the government is mandated to disclose publicly a roster of chemicals acknowledged for their ability to cause cancer or reproductive harm. In compliance with P65 guidelines, two categories of regulations have been established: No-Significant-Risk levels (NSRLs) for carcinogens and Maximum Allowable Dose levels (MADLs) for substances exhibiting reproductive or developmental toxicity [56]. The regulation of chemicals is contingent upon their effects on critical toxicological endpoints, particularly those related to reproductive or developmental toxicity and tumorigenesis. Benzene, DEHP, lead, methyl-mercury, nitrates, PAHs, and PCBs are among the chemicals that have been assessed, as previously discussed.
Conclusion and Future Direction
Biomarkers have been useful in studies of environmental chemical exposures and the associated causal inferences and risks. Their direct utilization in quantitative and qualitative risk assessment, as well as the development of chemical regulations, is somewhat constrained. The identification of biomarkers that can be studied throughout the entire developmental process necessitates tools and methodologies that are both feasible and cost-effective for larger sample sizes of patients. Nevertheless, the potential for expanded applications of biomarkers in these domains is promising. It is imperative to acknowledge that various factors can influence biomarker responses, including internal biological reactions, exposure to other chemicals, individual genetic profiles, gender, age, diet, physical activity, and pharmaceutical usage, among others. Comprehending these potential modifying factors is critical for effective risk assessments, as they can impact any stage of the sequential events. The presence of these influencing factors, as well as the diverse functions of different biomarkers, underscores the importance of integrating data from multiple biomarkers to enhance understanding of biological and toxicological processes, thereby enhancing risk assessments and creating a scientific foundation for environmental risk management and decision-making.
Ongoing research into validating biomarkers and leveraging high-throughput technologies will contribute to further advancements in biomarker development, risk assessment, and regulatory policy strategies. Furthermore, numerous existing and emerging tools are being specifically designed for use in both preclinical and clinical stages of drug discovery and development. Significantly, the transition of pharmacological compounds and medications from preclinical phases to clinical trials is substantially facilitated by methodologies capable of bridging these procedural segments.
- Kalali A, Richerson S, Ouzunova E, Westphal R, Miller B. Digital biomarkers in clinical drug development. in, Handbook of Behavioral Neuroscience. Elsevier. 2019; 229-238.
- Vorkamp K, Castaño A, Antignac JP, Boada LD, Cequier E, Covaci A, Esteban López M, Haug LS, Kasper-Sonnenberg M, Koch HM, Pérez Luzardo O, Osīte A, Rambaud L, Pinorini MT, Sabbioni G, Thomsen C. Biomarkers, matrices and analytical methods targeting human exposure to chemicals selected for a European human biomonitoring initiative. Environ Int. 2021 Jan;146:106082. doi: 10.1016/j.envint.2020.106082. Epub 2020 Nov 20. PMID: 33227583.
- Neuman MG. Biomarkers of Drug-Induced Liver Toxicity. Ther Drug Monit. 2019 Apr;41(2):227-234. doi: 10.1097/FTD.0000000000000610. PMID: 30883515.
- Bhutani P, Joshi G, Raja N, Bachhav N, Rajanna PK, Bhutani H, Paul AT, Kumar R. U.S. FDA Approved Drugs from 2015-June 2020: A Perspective. J Med Chem. 2021 Mar 11;64(5):2339-2381. doi: 10.1021/acs.jmedchem.0c01786. Epub 2021 Feb 22. PMID: 33617716.
- Kraus VB. Biomarkers as drug development tools: discovery, validation, qualification and use. Nat Rev Rheumatol. 2018 Jun;14(6):354-362. doi: 10.1038/s41584-018-0005-9. PMID: 29760435.
- Dalmolin SP, Dreon DB, Thiesen FV, Dallegrave E. Biomarkers of occupational exposure to pesticides: Systematic review of insecticides. Environ Toxicol Pharmacol. 2020 Apr;75:103304. doi: 10.1016/j.etap.2019.103304. Epub 2019 Nov 22. PMID: 31841723.
- Malekirad AA, Hassani S, Abdollahi M. Oxidative stress and copper smelter workers. in, Toxicology. Elsevier. 2021; 119-126.
- Sánchez-Alarcón J, Milić M, Kašuba V, Tenorio-Arvide MG, Montiel-González JMR, Bonassi S, Valencia-Quintana R. A Systematic Review of Studies on Genotoxicity and Related Biomarkers in Populations Exposed to Pesticides in Mexico. Toxics. 2021 Oct 21;9(11):272. doi: 10.3390/toxics9110272. PMID: 34822663; PMCID: PMC8624200.
- Kreth S, Hübner M, Hinske LC. MicroRNAs as Clinical Biomarkers and Therapeutic Tools in Perioperative Medicine. Anesth Analg. 2018 Feb;126(2):670-681. doi: 10.1213/ANE.0000000000002444. PMID: 28922229.
- Samad AFA, Kamaroddin MF. Innovative approaches in transforming microRNAs into therapeutic tools. Wiley Interdiscip Rev RNA. 2023 Jan;14(1):e1768. doi: 10.1002/wrna.1768. Epub 2022 Nov 27. PMID: 36437633.
- Hughes W. Essentials of environmental toxicology. CRC Press. 2019; 176.
- Aronson JK, Ferner RE. Biomarkers-A General Review. Curr Protoc Pharmacol. 2017 Mar 17;76:9.23.1-9.23.17. doi: 10.1002/cpph.19. PMID: 28306150.
- Organization WH. Biomarkers in risk assessment: Validity and validation. World Health Organization. 2001; 238.
- Hernández AF, Gil F, Tsatsakis AM. Biomarkers of chemical mixture toxicity. in, Biomarkers in toxicology. Elsevier. 2019; 569-585.
- Li Y, Wang X, Feary McKenzie J, 't Mannetje A, Cheng S, He C, Leathem J, Pearce N, Sunyer J, Eskenazi B, Yeh R, Aylward LL, Donovan G, Mueller JF, Douwes J. Pesticide exposure in New Zealand school-aged children: Urinary concentrations of biomarkers and assessment of determinants. Environ Int. 2022 May;163:107206. doi: 10.1016/j.envint.2022.107206. Epub 2022 Apr 5. PMID: 35395578.
- Robson MG, Toscano WA. Toxicological Basis for Risk Assessment. in, Risk Assessment for Environmental Health. CRC Press. 2022; 111-151.
- Timbrell J, Barile FA. Introduction to toxicology. CRC Press. 2023; 316.
- Robson MG, Toscano WA, Meng Q, Kaden DA. Risk assessment for environmental health. CRC Press. 2022; 400.
- Clewell RA, Thompson CM, Clewell HJ 3rd. Dose-dependence of chemical carcinogenicity: Biological mechanisms for thresholds and implications for risk assessment. Chem Biol Interact. 2019 Mar 1;301:112-127. doi: 10.1016/j.cbi.2019.01.025. Epub 2019 Feb 11. PMID: 30763550.
- Grandjean P. Biomarkers in epidemiology. Clin Chem. 1995 Dec;41(12 Pt 2):1800-3. PMID: 7497635.
- Hall AL, MacLean MB, VanTil L, McBride DI, Glass DC. Considering Exposure Assessment in Epidemiological Studies of Chronic Health in Military Populations. Front Public Health. 2020 Oct 6;8:577601. doi: 10.3389/fpubh.2020.577601. PMID: 33123510; PMCID: PMC7573167.
- Vidal A, Mengelers M, Yang S, De Saeger S, De Boevre M. Mycotoxin Biomarkers of Exposure: A Comprehensive Review. Compr Rev Food Sci Food Saf. 2018 Sep;17(5):1127-1155. doi: 10.1111/1541-4337.12367. Epub 2018 Jul 6. PMID: 33350155.
- Watson WP, Mutti A. Role of biomarkers in monitoring exposures to chemicals: present position, future prospects. Biomarkers. 2004 May-Jun;9(3):211-42. doi: 10.1080/13547500400015642. PMID: 15764289.
- Assis CRD, Linhares AG, Cabrera MP, Oliveira VM, Silva KCC, Marcuschi M, Maciel Carvalho EVM, Bezerra RS, Carvalho LB Jr. Erythrocyte acetylcholinesterase as biomarker of pesticide exposure: new and forgotten insights. Environ Sci Pollut Res Int. 2018 Jul;25(19):18364-18376. doi: 10.1007/s11356-018-2303-9. Epub 2018 May 24. PMID: 29797194.
- Gupta RC. Biomarkers in toxicology. Academic press. 2019; 1246.
- Sarhadi VK, Armengol G. Molecular Biomarkers in Cancer. Biomolecules. 2022 Jul 23;12(8):1021. doi: 10.3390/biom12081021. PMID: 35892331; PMCID: PMC9331210.
- Hwa Yun B, Guo J, Bellamri M, Turesky RJ. DNA adducts: Formation, biological effects, and new biospecimens for mass spectrometric measurements in humans. Mass Spectrom Rev. 2020 Mar;39(1-2):55-82. doi: 10.1002/mas.21570. Epub 2018 Jun 11. PMID: 29889312; PMCID: PMC6289887.
- Menz J, Götz ME, Gündel U, Gürtler R, Herrmann K, Hessel-Pras S, Kneuer C, Kolrep F, Nitzsche D, Pabel U, Sachse B, Schmeisser S, Schumacher DM, Schwerdtle T, Tralau T, Zellmer S, Schäfer B. Genotoxicity assessment: opportunities, challenges and perspectives for quantitative evaluations of dose-response data. Arch Toxicol. 2023 Sep;97(9):2303-2328. doi: 10.1007/s00204-023-03553-w. Epub 2023 Jul 5. PMID: 37402810; PMCID: PMC10404208.
- Thomas CE, Wang R, Adams-Haduch J, Murphy SE, Ueland PM, Midttun Ø, Brennan P, Johansson M, Gao YT, Yuan JM. Urinary Cotinine Is as Good a Biomarker as Serum Cotinine for Cigarette Smoking Exposure and Lung Cancer Risk Prediction. Cancer Epidemiol Biomarkers Prev. 2020 Jan;29(1):127-132. doi: 10.1158/1055-9965.EPI-19-0653. Epub 2019 Nov 4. PMID: 31685561; PMCID: PMC7695222.
- Ma B, Stepanov I, Hecht SS. Recent Studies on DNA Adducts Resulting from Human Exposure to Tobacco Smoke. Toxics. 2019 Mar 19;7(1):16. doi: 10.3390/toxics7010016. PMID: 30893918; PMCID: PMC6468371.
- Rogers S, de Souza AR, Zago M, Iu M, Guerrina N, Gomez A, Matthews J, Baglole CJ. Aryl hydrocarbon receptor (AhR)-dependent regulation of pulmonary miRNA by chronic cigarette smoke exposure. Sci Rep. 2017 Jan 12;7:40539. doi: 10.1038/srep40539. PMID: 28079158; PMCID: PMC5227990.
- Fan AM. Biomarkers in toxicology, risk assessment, and environmental chemical regulations. in, Biomarkers in toxicology. Elsevier. 2014; 1057-1080.
- Alvarez CS, Hernández E, Escobar K, Villagrán CI, Kroker-Lobos MF, Rivera-Andrade A, Smith JW, Egner PA, Lazo M, Freedman ND, Guallar E, Dean M, Graubard BI, Groopman JD, Ramírez-Zea M, McGlynn KA. Aflatoxin B1 exposure and liver cirrhosis in Guatemala: a case-control study. BMJ Open Gastroenterol. 2020 Jul;7(1):e000380. doi: 10.1136/bmjgast-2020-000380. PMID: 32641287; PMCID: PMC7342465.
- Marín S, Ramos AJ, Sanchis V, Cano-Sancho G. An overview of mycotoxin biomarker application in exposome-health studies. Current Opinion in Food Science. 2021; 39:31-35.
- Farmer PB. Biomonitoring and molecular dosimetry of genotoxic carcinogens. in, Molecular and Cellular Mechanisms of Toxicity. CRC Press. 2019; 101-124.
- Smith J, Groopman J. Translating DNA Adduct Knowledge to Human Prevention Trials. in, DNA Damage, DNA Repair and Disease. 2020; 361-387.
- Haschek WM, Rousseaux CG, Wallig MA, Bolon B. Haschek and Rousseaux's Handbook of Toxicologic Pathology, Volume 3: Environmental Toxicologic Pathology and Major Toxicant Classes. Elsevier. 2023; 1152.
- Baugh EH, Ke H, Levine AJ, Bonneau RA, Chan CS. Why are there hotspot mutations in the TP53 gene in human cancers? Cell Death Differ. 2018 Jan;25(1):154-160. doi: 10.1038/cdd.2017.180. Epub 2017 Nov 3. PMID: 29099487; PMCID: PMC5729536.
- Zakharia K, Luther CA, Alsabbak H, Roberts LR. Hepatocellular carcinoma: Epidemiology, pathogenesis and surveillance - implications for sub-Saharan Africa. S Afr Med J. 2018 Aug 8;108(8b):35-40. doi: 10.7196/SAMJ.2018.v108i8b.13499. PMID: 30182911.
- Wang H, Chen L, Zhou T, Zhang Z, Zeng C. p53 Mutation at Serine 249 and Its Gain of Function Are Highly Related to Hepatocellular Carcinoma after Smoking Exposure. Public Health Genomics. 2021;24(3-4):171-181. doi: 10.1159/000516598. Epub 2021 Jun 30. PMID: 34192689.
- Long XD, Blumenberg M. Aflatoxin B1 Occurrence, Detection and Toxicological Effects. IntechOpen. 2020 220.
- Stornetta A, Zimmermann M, Cimino GD, Henderson PT, Sturla SJ. DNA Adducts from Anticancer Drugs as Candidate Predictive Markers for Precision Medicine. Chem Res Toxicol. 2017 Jan 17;30(1):388-409. doi: 10.1021/acs.chemrestox.6b00380. Epub 2017 Jan 3. PMID: 27936622; PMCID: PMC5379252.
- Anna A, Monika G. Splicing mutations in human genetic disorders: examples, detection, and confirmation. J Appl Genet. 2018 Aug;59(3):253-268. doi: 10.1007/s13353-018-0444-7. Epub 2018 Apr 21. Erratum in: J Appl Genet. 2019 May;60(2):231. PMID: 29680930; PMCID: PMC6060985.
- Usman MB, Priya K, Pandit S, Gupta PK, Agrawal S, Sarma H, Prasad R. Genetic polymorphisms and Pesticide-Induced DNA damage: a review. The Open Biotechnology Journal. 2021; 15(1).
- Kali Z, Cagiran FT, Kirici P, Dogan C, Celik O. DNA repair gene (XRCC1 and XPD) polymorphism and risk of primary ovarian failure. Eur Rev Med Pharmacol Sci. 2022 Sep;26(18):6593-6599. doi: 10.26355/eurrev_202209_29759. PMID: 36196709.
- Moffett DB, Mumtaz MM, Sullivan Jr DW, Whittaker MH. General considerations of dose-effect and dose-response relationships. in, Handbook on the Toxicology of Metals. Elsevier. 2022; 299-317.
- Wilson-Frank C. Proteomics in Biomarkers of Chemical Toxicity. in, Biomarkers in Toxicology. Elsevier. 2019; 1153-1163.
- Silins I, Högberg J. Combined toxic exposures and human health: biomarkers of exposure and effect. Int J Environ Res Public Health. 2011 Mar;8(3):629-47. doi: 10.3390/ijerph8030629. Epub 2011 Feb 24. PMID: 21556171; PMCID: PMC3083662.
- Jaladanki CK, Gahlawat A, Rathod G, Sandhu H, Jahan K, Bharatam PV. Mechanistic studies on the drug metabolism and toxicity originating from cytochromes P450. Drug Metab Rev. 2020 Aug;52(3):366-394. doi: 10.1080/03602532.2020.1765792. Epub 2020 Jul 9. PMID: 32645272.
- Santonen T, Aitio A, Fowler BA, Nordberg M. Biological monitoring and biomarkers. in, Handbook on the Toxicology of Metals. Elsevier. 2015; 155-171.
- Bonassi S, Neri M, Puntoni R. Validation of biomarkers as early predictors of disease. Mutat Res. 2001 Sep 1;480-481:349-58. doi: 10.1016/s0027-5107(01)00194-4. PMID: 11506827.
- Janasie C, Deans O, Harris T. Legal Extension Strategies to Increase Awareness of Drinking Water Contaminant Regulatory Framework. Journal of Contemporary Water Research & Education. 2021; 174(1):106-119.
- Dozier M, McFarland ML. Drinking water Standards. Texas FARMER Collection. 2006.
- Azuma K, Jinno H, Tanaka-Kagawa T, Sakai S. Risk assessment concepts and approaches for indoor air chemicals in Japan. Int J Hyg Environ Health. 2020 Apr;225:113470. doi: 10.1016/j.ijheh.2020.113470. Epub 2020 Feb 9. PMID: 32050149.
- Wollin KM, Apel P, Chovolou Y, Pabel U, Schettgen T, Kolossa-Gehring M, Röhl C, Agency OBOTHBCOTGE. Concept for the Evaluation of Carcinogenic Substances in Population-Based Human Biomonitoring. Int J Environ Res Public Health. 2022 Jun 13;19(12):7235. doi: 10.3390/ijerph19127235. PMID: 35742488; PMCID: PMC9223427.
- Luz AL, Wu X, Tokar EJ. Toxicology of inorganic carcinogens. in, Advances in Molecular Toxicology. Elsevier. 2018; 1-46.
Article Alerts
Subscribe to our articles alerts and stay tuned.
 This work is licensed under a Creative Commons Attribution 4.0 International License.
This work is licensed under a Creative Commons Attribution 4.0 International License.


 Save to Mendeley
Save to Mendeley
